Frequently Asked Questions (FAQ):
1. Question: What is an appropriate cell seeding density?
This should be determined through preliminary experiments. Typically, for a 96-well plate, 3,000 to 7,000 cells are seeded per well (adjusted according to the cell growth rate) to ensure that, at the end of the culture period, the cells in the control wells have just reached full confluence or are nearly confluent (approximately 80–90% confluence).
2. Question: Will poor cell condition affect the results?
It will have a significant impact. Ensure that the cells used for the experiment are in the logarithmic growth phase, possess high viability, and are in a stable condition; avoid using cells that have undergone too many passages.
3. Question: What should be done if a large number of adherent cells detach during the experiment?
Possible causes include: excessive trypsin digestion (leading to the disruption of intercellular junctions); insufficient serum concentration in the culture medium; or poor intrinsic cell condition (e.g., failure to fully recover after thawing).
Solutions: 1. Monitor the trypsin digestion process under a microscope; as soon as the intercellular spaces widen and the cell edges become rounded, immediately add serum-containing medium to terminate the digestion. 2. Adjust the serum concentration in the culture medium to 10–15%. 3. After thawing, culture the cells for 48 hours to allow them to fully adhere and spread before proceeding with drug treatment.
4. Question: Can cells continue to be cultured after adding the CCK-8 reagent?
Yes, but the following steps are required: Carefully aspirate the culture medium containing the CCK-8 reagent and the orange-yellow formazan product (be careful not to aspirate the cells!). Gently rinse the wells 2–3 times with pre-warmed PBS to thoroughly wash away any residual reagent. Then, add complete culture medium and return the plate to the incubator.
5. Question: Why do cells appear morphologically normal after drug treatment, yet exhibit low OD values?
The drug inhibits the activity of cellular metabolic enzymes (without causing cell death); competitive inhibition occurs between the CCK-8 reagent and the drug; or the cells have entered a dormant phase.
Solutions: 1. Verify cell viability using a live/dead cell staining kit (e.g., Calcein-AM/PI) to rule out interference from metabolic inhibition. 2. Include a "Drug + CCK-8 + Culture Medium" blank control group to subtract the drug's influence on color development. 3. Extend the recovery period following drug treatment (e.g., by culturing for an additional 12 hours) before measuring the OD values.
6. Question: Why do "plaques" (localized cell-free areas) appear after cell seeding?
Air bubbles are present in the cell suspension (introduced during pipetting); impurities are present at the bottom of the culture plate wells (due to insufficient cleaning); or the cell suspension concentration is uneven.
Solutions: 1. Inspect the cell suspension before pipetting; if bubbles are present, use a pipette tip to puncture and eliminate them. 2. Rinse the culture plates twice with ultrapure water before use, and allow them to air-dry completely. 3. Thoroughly mix the cell suspension before aliquoting, and re-mix it after dispensing every 10 wells.
7. Question: What should be done if the cell mortality rate is extremely high and no inhibitory effect is observed?
Reduce the drug concentration or shorten the treatment duration; alternatively, verify whether the toxicity is caused by the solvent (e.g., DMSO).
8. Question: Can CCK-8 be used with bacterial or fungal cells?
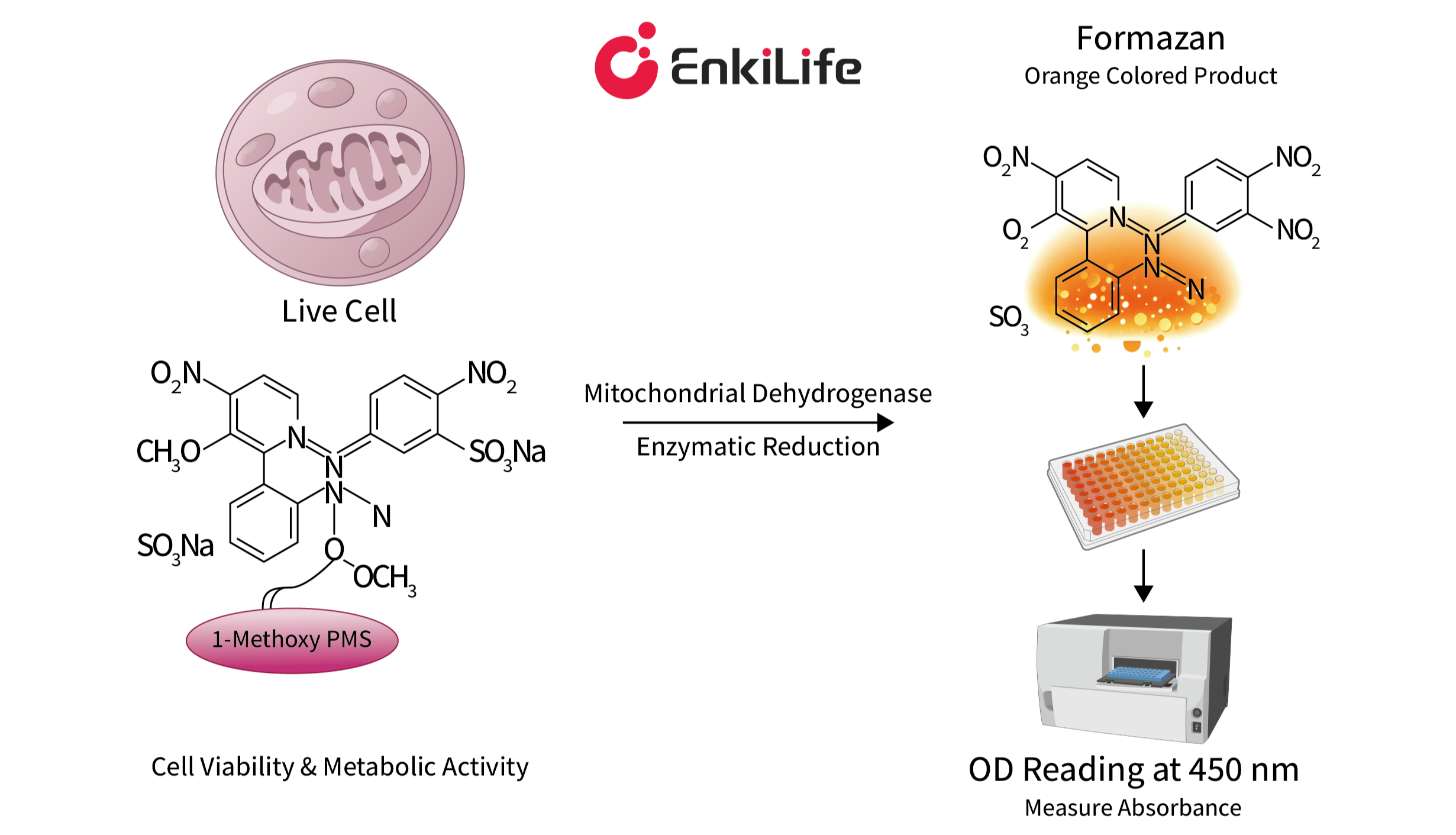
Solution: No. The working principle of CCK-8 relies on the dehydrogenase enzymes located within the mitochondria of eukaryotic cells.
9. Question: At what stage of cell culture should the drug be added?
Solution: Adherent cells undergo four distinct stages after seeding: the "suspension phase" (0–6 h), the "attachment phase" (6–12 h), the "spreading phase" (12–24 h), and the "proliferation phase" (after 24 h). The true effects of a drug can only be accurately reflected once the cells have completed attachment and spreading, and have entered the proliferation phase. Therefore, under standard experimental conditions, it is recommended to add the drug 24–48 hours after seeding. Unlike adherent cells, suspension cells do not require a waiting period to attach and adapt. Provided the seeding density is appropriate, they typically enter the logarithmic growth phase—characterized by vigorous metabolism, stable proliferation, and high sensitivity to drug responses—within 12–24 hours post-seeding. Therefore, under standard experimental conditions, it is recommended to add test compounds within 12–24 hours after seeding.
10. Question: How long is the incubation period for the CCK-8 assay?
Typically, the incubation period is 1–4 hours. The optimal incubation time should be determined through preliminary experiments; the linear correlation is strongest when the OD values fall within the range of 1.0 to 2.0.
Find out more here:Comprehensive Guide to CCK-8 Assays: From Principle to Practice——Efficiently Obtaining Reliable Cell Proliferation Data