HSP/Chaperone Antibody
HSP70 and HSP90 are molecular chaperones expressed constitutively under normal conditions to maintain protein homeostasis and are induced upon environmental stress (1). HSP70 and HSP90 interact with unfolded proteins to prevent irreversible aggregation and catalyze the refolding of their substrates in an ATP-dependent manner (1). HSP40 family proteins bind unfolded proteins and prevent their aggregation, and deliver unfolded protiens to HSP70 (2). HSP60 has primarily been known as a mitochondrial protein that is important for folding key proteins after import into the mitochondria (3). HSP60 is also present in the cytosol of many cells and is induced by stress, inflammatory and immune responses, autoantibodies correlated with Alzheimer's, coronary artery diseases, MS, and diabetes (4-7). Secretory and transmembrane proteins are synthesized on polysomes and translocate into the endoplasmic reticulum (ER) where they are often modified by the formation of disulfide bonds, amino-linked glycosylation and folding. The ER contains a pool of molecular chaperones including calnexin, BiP and protein disulfide isomerase (PDI). Calenxin is a calcium-binding protein embedded in the ER membrane that retains newly synthesized glycoproteins inside the ER to ensure proper folding and quality control (8,9). When protein folding is disturbed inside the ER, Bip synthesis is increased. Subsequently, BiP binds to misfolded proteins to prevent them from forming aggregates and assists them to refold properly (10). PDI catalyzes the formation and isomerization of disulfide bonds required to reach a proteins native state (11). Heat shock gene transcription is regulated by a familly of heat shock factors (HSFs), transcriptional activators that bind to heat shock response elements (HSEs) located upstream of all heat shock genes (12).
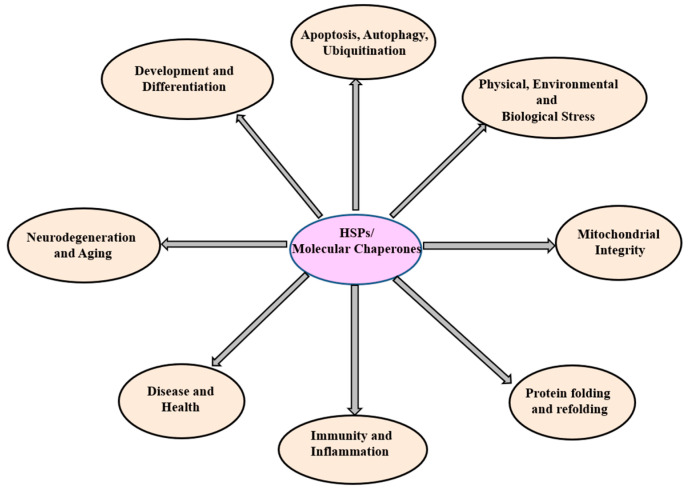
The diagram illustrates HSPs and their associated functions. HSPs (HSP100, HSP90, HSP70, HSP60, SHSPs, ubiquitin) oversee diverse cellular functions.
Relevant Antibodies
| Catalog# | Product Name | Application | Reactivity |
|---|---|---|---|
| AMRe21510 | Hsp60 Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| AMRe02126 | Hsp70 Rabbit Monoclonal Antibody | WB,IHC-P | Human |
| AMRe12221 | HSF1 (14S18) Rabbit Monoclonal Antibody | WB,IHC-P,ICC/IF,FC,IP,IF-P | Human |
| AMRe21472 | GRP78/BiP Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| AMM03613 | Hsp40 Mouse Monoclonal Antibody | WB | Human |
| AMRe21616 | Hsp90 Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| AMRe01749 | Calnexin Rabbit Monoclonal Antibody | WB | Human |
| AMRe21290 | PDI Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| APS0635 | HRP-conjugated Polyclonal Goat Anti-Rabbit IgG(H+L) Secondary Antibody | ELISA,WB,Dotblot | Mouse |
| APS0631 | HRP-conjugated Polyclonal Goat Anti-Mouse IgG(H+L) Secondary Antibody | ELISA,WB,Dotblot | Mouse |
| AMRe80004 | GAPDH (12R9) Rabbit Monoclonal Antibody | WB,ELISA | Human,Mouse,Rat,Rabbit,Dog,Monkey |
Related Products
Super-sensitive ECL chemiluminescent reagent
References
- Nollen, E.A. and Morimoto, R.I. (2002) J. Cell Sci. 115, 2809-2816.
- Fan, C.Y. et al. (2003) Cell Stress Chaperones 8, 309-316.
- Jindal, S. et al. (1989) Mol Cell Biol 9, 2279-83.
- Itoh, H. et al. (2002) Eur. J. Biochem. 269, 5931-5938.
- Gupta, S. and Knowlton, A.A. J. Cell Mol. Med. 9, 51-58.
- Deocaris, C.C. et al. (2006) Cell Stress Chaperones 11, 116-128.
- Lai, H.C. et al. (2007) Am. J. Physiol. Endocrinol. Metab. 292, E292-E297.
- Bergeron, J.J. et al. (1994) Trends Biochem. Sci. 19, 124-128.
- Williams, D.B. (2006) J. Cell Sci. 119, 615-623.
- Kohno, K. et al. (1993) Mol. Cell. Biol. 13, 877-890.
- Ellgaard, L. and Ruddock, L.W. (2005) EMBO Rep. 6, 28-32.
- Morimoto, R.I. (1998) Genes Dev. 12, 3788-3796.
- Satyal, S.H. et al. (1998) Genes Dev. 12, 1962-1974.
- Singh MK, et al (2024) Int J Mol Sci. Apr 10;25(8):4209.
 | Voisey Voisey is a technical support specialist at EnkiLife, proficient in immunology and cell biology. She is committed to providing customers with professional and efficient technical support. Additionally, she is involved in research on customers' fields of study and designs highly cost-effective solutions for them. |
