Glutamine Metabolism
ASCT2 (SLC1A5)
Sodium-dependent neutral amino acid transporter type 2 (ASCT2 or SLC1A5) is a neutral amino acid transporter that regulates the uptake of essential amino acids in conjunction with the SLC7A5 bilateral transporter. ASCT2 appears to be the major glutamine transporter in hepatoma cells and is thought to provide essential amino acids needed for tumor growth. Additional evidence suggests that ASCT2 plays a role in activating mTORC1 signaling and is required to suppress autophagy. Cell surface ASCT2 serves as a receptor for several mammalian interference retroviruses associated with cases of infectious immunodeficiency; variation in a small region of an extracellular loop (ECL2) may be responsible for species-specific differences in receptor function.
GFAT1 & GFAT2
GFAT1, glutamine:fructose-6-phosphate aminotransferase 1, is the rate-limiting enzyme of the hexosamine biosynthesis pathway. This enzyme catalyzes the conversion of fructose-6-phosphate and glutamine to glucosamine-6-phosphate and glutamate. The hexosamine biosynthesis pathway generates the building blocks for protein and lipid glycosylation. Furthermore, studies suggest that increased activity of this pathway is a contributing factor to hyperglycemia-induced insulin resistance. GFAT1 is more active in non-insulin-dependent diabetes mellitus (NIDDM) patients. Transgenic mice overexpressing this enzyme in skeletal muscle and adipose tissue show an insulin resistance phenotype.
GFAT2, an isoenzyme of GFAT1, was later identified. Studies show that the regulation of GFAT2 is different from that of GFAT1, suggesting differential regulation of the hexosamine pathway in different tissues.
Glutaminase (GLS1 & GLS2)
Glutaminase catalyzes the conversion of glutamine to glutamate, the first and rate-limiting step of glutaminolysis. Both kidney-type glutaminase (GLS1) and liver-type glutaminase (GLS2) are found in mammals. GLS1-mediated glutathione synthesis plays an essential role in redox homeostasis and contributes to increased survival of postimplantation bone cells preconditioned to the hypoxic and ischemic environment in the bone defect site. In addition, KEAP1–NRF2-mutant LUAD (KRAS-mutant lung adenocarcinoma) tumors are dependent on increased glutaminolysis. Furthermore, recent studies showed higher glutaminolysis and glucose production from glutamine in human primary hepatocytes with GLS2 gain-of-function missense mutations. These findings suggest GLS1 and GLS2 as potential targets in the therapy of bone regeneration and in the treatments of diseases such as cancer and hyperglycemia, respectively.
Glutamate Dehydrogenase
Glutamate dehydrogenase is a mitochondrial enzyme that catalyzes the oxidative deamination of glutamate to α-ketoglutarate through association with the cofactor nicotinamide adenine dinucleotide phosphate. Glutamate dehydrogenase is highly expressed in various tissues such as the liver, brain, kidney, heart, pancreas, ovaries, and testis. Two isoforms produced by two distinct genes are found in mammalian tissues. The GLUD1 gene is ubiquitously expressed, while the GLUD2 gene is specifically expressed in testicular tissues and astrocytes. Glutamate dehydrogenase links glutamate to the Krebs cycle, thereby playing a critical role in the regulation of energy homeostasis. Research studies have shown that changes in glutamate dehydrogenase activity in pancreatic β-cells can cause a hyperinsulinism syndrome.
GOT1 & ASNS
Glutamate oxaloacetate transaminase 1 (GOT1) catalyzes the interconversion of aspartate and oxaloacetate.
Asparagine synthetase (ASNS) catalyzes the synthesis of asparagine from aspartate and glutamine. In subsets of gastric and hepatic cancers, ASNS promoter hypermethylation correlates with low ASNS expression, sensitizing these cancers to the asparaginase treatment.
Glutamine Metabolism Pathway Diagram
PURINES → GLUTAMINE → PYRIMIDINES / NAD / CO₂ / GLUCOSAMINE / ASPARAGINE / GLUTAMATE → GLUTATHIONE / PROLINE / ORNITHINE / ARGININE / GLUCOSE / AMMONIA / UREA
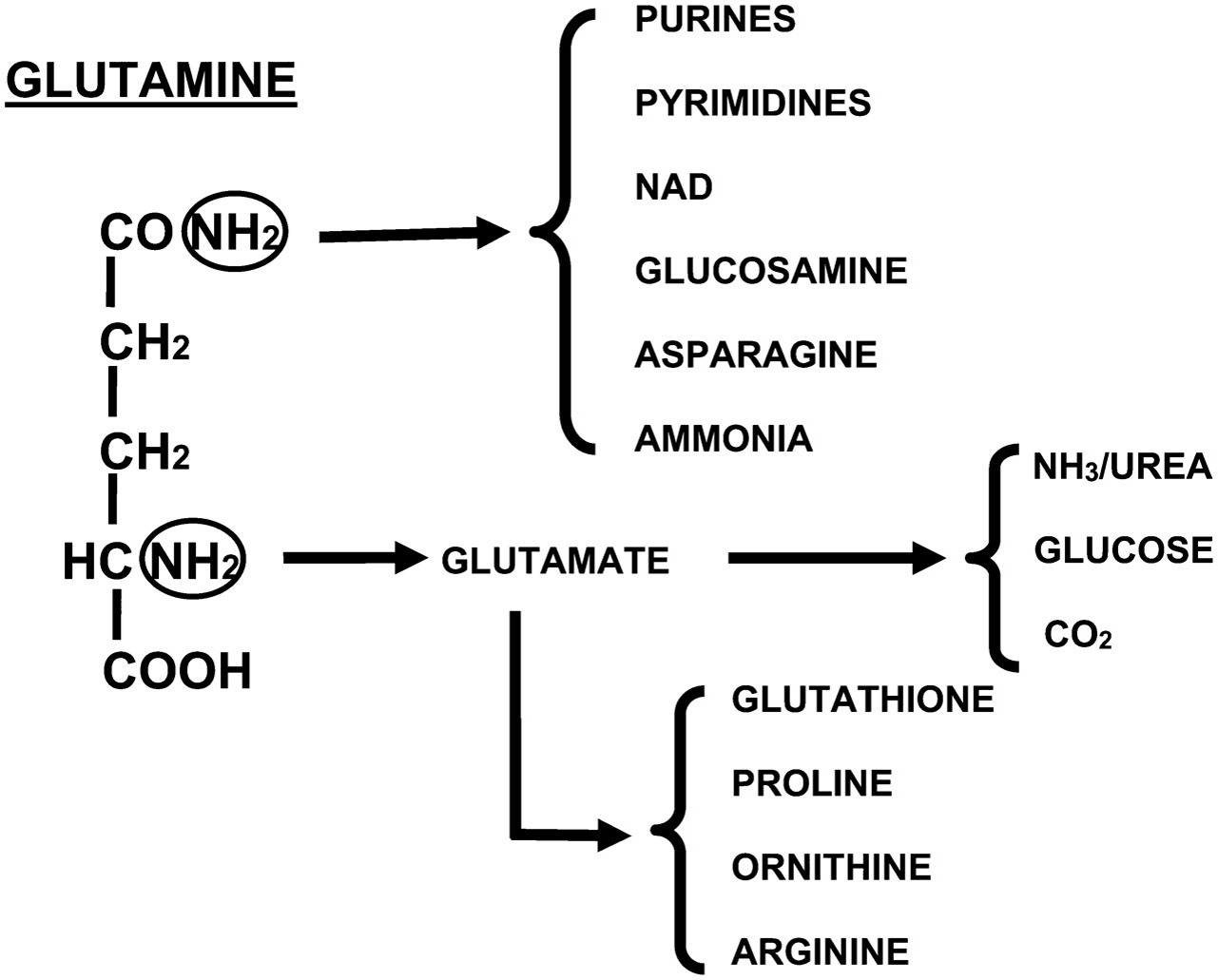 Glutamine Metabolism Pathway Diagram
Glutamine Metabolism Pathway Diagram
Relevant Antibodies
| Catalog# | Product Name | Reactivity | Application |
|---|---|---|---|
| AMRe84275 | FANCA Rabbit Monoclonal | Human | WB, IP |
| AMRe01974 | FANCB Rabbit Monoclonal Antibody | Human, Mouse | WB |
| AMRe10826 | FANCD2 (16Q3) Rabbit Monoclonal Antibody | Human, Mouse, Rat | WB, IHC-P, ICC/IF, FC, IP, IF-P |
| AMM86025 | BRCA2 Mouse Monoclonal Antibody | Human | WB |
| AMM82390 | BRIP1 Mouse Monoclonal Antibody | Human | IHC, FC, ELISA |
| APS0635 | HRP-conjugated Polyclonal Goat Anti-Rabbit IgG(H+L) Secondary Antibody | Rabbit | ELISA, WB, Dot blot |
| AMre80004 | GAPDH (12R9) Rabbit Monoclonal Antibody | Human, Mouse, Rat, Rabbit, Dog, Monkey | WB, ELISA |
| AMRe01444 | Asparagine Synthetase Rabbit Monoclonal Antibody | Human, Rat | WB, IHC-P |
| APS0631 | HRP-conjugated Polyclonal Goat Anti-Mouse IgG(H+L) Secondary Antibody | — | — |
| APS0635 | HRP-conjugated Polyclonal Goat Anti-Rabbit IgG(H+L) Secondary Antibody | — | — |
Related Products
Super-sensitive ECL chemiluminescent reagent
References
Utsunomiya-Tate N, Endou H, Kanai Y. Cloning and functional characterization of a system ASC-like Na+-dependent neutral amino acid transporter. J Biol Chem. 1996 Jun 21;271(25):14883-90. [PMID: 8662767].
Oki T, Yamazaki K, Kuromitsu J, Okada M, Tanaka I. cDNA cloning and mapping of a novel subtype of glutamine:fructose-6-phosphate amidotransferase (GFAT2) in human and mouse. Genomics. 1999 Apr 15;57(2):227-34.[PMID: 10198162].
Aledo JC, Gómez-Fabre PM, Olalla L, Márquez J. Identification of two human glutaminase loci and tissue-specific expression of the two related genes. Mamm Genome. 2000 Dec;11(12):1107-10. [PMID: 11130979].
Zaganas I, Spanaki C, Plaitakis A. Expression of human GLUD2 glutamate dehydrogenase in human tissues: functional implications. Neurochem Int. 2012 Sep;61(4):455-62[PMID: 22709674].
Li H, Ning S, Ghandi M, Kryukov GV, Gopal S, Deik A, Souza A, Pierce K, Keskula P, Hernandez D, Ann J, Shkoza D, Apfel V, Zou Y, Vazquez F, Barretina J, Pagliarini RA, Galli GG, Root DE, Hahn WC, Tsherniak A, Giannakis M, Schreiber SL, Clish CB, Garraway LA, Sellers WR. The landscape of cancer cell line metabolism. Nat Med. 2019 May;25(5):850-860.[PMID: 31068703].
Watford M. Glutamine metabolism and function in relation to proline synthesis and the safety of glutamine and proline supplementation. J Nutr. 2008 Oct;138(10):2003S-2007S.[PMID: 18806115].
 | Flora Flora is a technical support expert at EnkiLife, familiar with immunology and neuroscience, dedicated to providing customers with high-quality product combinations and technical support to help achieve research in neurodegenerative diseases and other neuroscience areas. |
