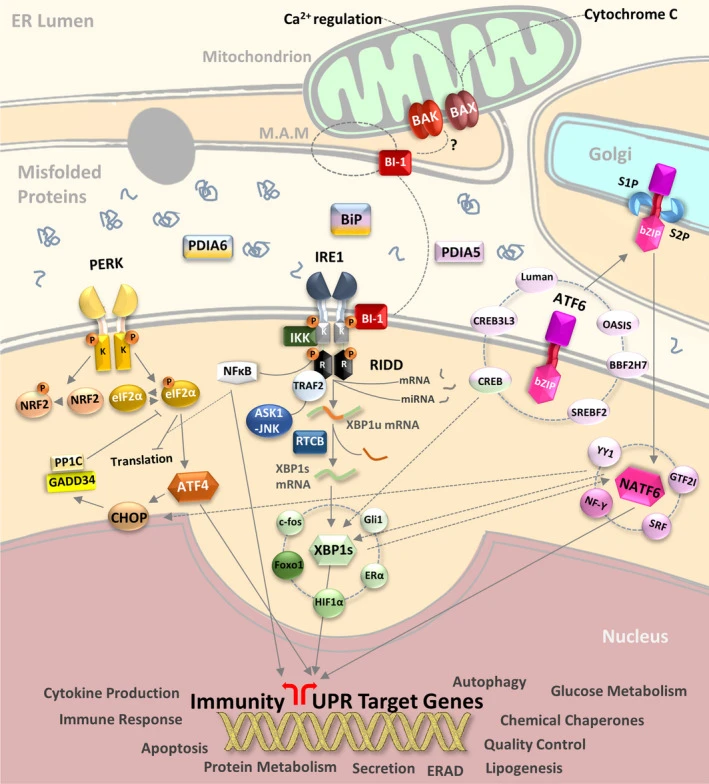
Secretory and transmembrane proteins are synthesized on polysomes and translocate into the endoplasmic reticulum (ER) where they are often modified by the formation of disulfide bonds, amino-linked glycosylation and folding. The ER contains a pool of molecular chaperone proteins including calnexin, BiP and protein disulfide isomerase (PDI). Calnexin is an ER membrane, calcium-binding protein that retains newly synthesized glycoproteins inside the ER to ensure proper folding and quality control (1,2). Irregular protein folding within the ER increases BiP synthesis, which binds misfolded proteins to prevent them from forming aggregates and to assist them to refold properly (3).
PDI catalyzes the formation and isomerization of disulfide bonds required for a protein to reach its native state (4). Studies have found that the resident ER protein endoplasmic oxidoreductin-1 (Ero1) provides oxidizing potential to the ER in Saccharomyces cerevisiae (5). Ero1-Lα is an ER membrane-associated N-glycoprotein that promotes oxidative protein folding (6). Disruptions of ER homeostasis leads to the accumulation of unfolded proteins. The ER has developed an adaptive mechanism called the unfolded protein response (UPR) to counteract compromised protein folding (7). This is regulated by proteins such as the membrane-bound transcription factor protease site 2 (MBTPS2) and the serine/threonine kinase IRE1 (8-12). The PERK eIF2α kinase is an ER resident transmembrane protein that couples ER stress signals to translation inhibition. ER stress increases PERK activity, which phosphorylates eIF2α to reduce protein translation. PERK activation during ER stress correlates with autophosphorylation of its cytoplasmic kinase domain (13,14). Phosphorylation of PERK at Thr980 can serve as a marker for its activation status.
During ER stress, the level of CHOP expression is elevated and CHOP functions to mediate programmed cell death (15).
| Catalog# | Product Name | Application | Reactivity |
|---|---|---|---|
| AMRe21472 | GRP78/BiP Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| AMRe01749 | Calnexin Rabbit Monoclonal Antibody | WB | Human |
| AMRe05925 | Phospho-IRE1 (S724) (4K17) Rabbit Monoclonal Antibody | WB | Human |
| AMRe21290 | PDI Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| AMRe21427 | CHOP Rabbit Monoclonal antibody | WB,IHC,IF,IP,ELISA | Human,Mouse,Rat |
| AMRe21506 | PERK Rabbit Monoclonal antibody | WB,IF,IP,ELISA | Human,Mouse,Rat |
| APS0635 | HRP-conjugated Polyclonal Goat Anti-Rabbit IgG(H+L) Secondary Antibody | ELISA,WB,Dotblot | Mouse |
| AMRe80004 | GAPDH (12R9) Rabbit Monoclonal Antibody | WB,ELISA | Human,Mouse,Rat,Rabbit,Dog,Monkey |
- Bergeron, J.J. et al. (1994) Trends Biochem. Sci. 19, 124-128.
- Williams, D.B. (2006) J. Cell Sci. 119, 615-623.
- Kohno, K. et al. (1993) Mol. Cell. Biol. 13, 877-890.
- Ellgaard, L. and Ruddock, L.W. (2005) EMBO Rep. 6, 28-32.
- Frand, A.R. and Kaiser, C.A. (1998) Mol. Cell 1, 161-170.
- Cabibbo, A. et al. (2000) J. Biol. Chem. 275, 4827-4833.
- Kaufman, R.J. et al. (2002) Nat. Rev. Mol. Cell Biol. 3, 411-421.
- Nikawa, J. and Yamashita, S. (1992) Mol. Microbiol. 6, 1441-1446.
- Cox, J.S. et al. (1993) Cell 73, 1197-1206.
- Mori, K. et al. (1993) Cell 74, 743-756.
- Lee, K. et al. (2002) Genes Dev. 16, 452-466.
- Shen, J. and Prywes, R. (2004) J. Biol. Chem. 279, 43046-43051.
- Harding, H.P. et al. (1999) Nature 397, 271-274.
- Shi, Y. et al. (1998) Mol. Cell. Biol. 18, 7499-7509.
- Zinszner, H. et al. (1998) Genes Dev 12, 982-95.
 | Voisey Voisey is a technical support specialist at EnkiLife, proficient in immunology and cell biology. She is committed to providing customers with professional and efficient technical support. Additionally, she is involved in research on customers' fields of study and designs highly cost-effective solutions for them. |
