Mitochondrial Membrane Potential Detection
Introduction to Mitochondrial Membrane Potential
Mitochondria are the main site for ATP production in animal and plant cells, and are important organelles that promote cellular energy conversion and participate in cell apoptosis. Mitochondria store the energy generated during respiratory oxidation in the inner membrane of mitochondria as electrochemical potential energy, causing asymmetric distribution of proton and other ion concentrations on both sides of the inner membrane and forming the mitochondrial membrane potential (MMP, Δ PSI m). Normal MMP is a prerequisite for maintaining mitochondrial oxidative phosphorylation and producing adenosine triphosphate. The stability of MMP is beneficial for maintaining normal physiological functions of cells.
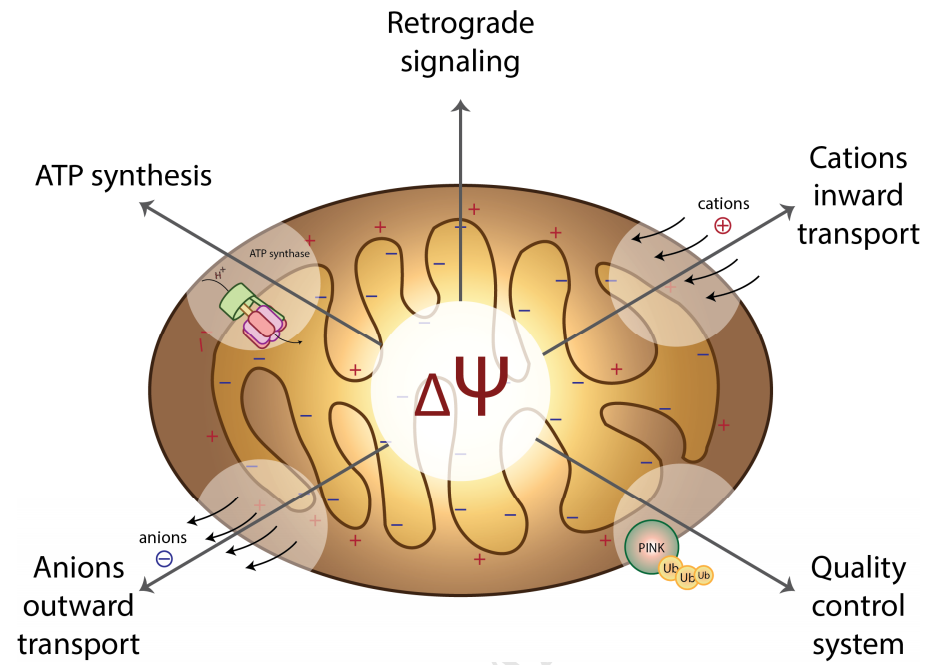
Figure 1: Mitochondrial Membrane Potential (Zorova et al., 2017)
Why Measure Mitochondrial Membrane Potential?
Mitochondria are important regulators of cellular energy and metabolism, playing a crucial role in maintaining cell growth and survival. The core function of mitochondria is to synthesize ATP through oxidative phosphorylation, which is well-known in mitochondrial bioenergetics. The production of ATP is related to mitochondrial membrane potential. Mitochondria maintain oxidative phosphorylation and drive ATP synthesis through membrane potential gradients generated by electron transport chains. This step is accomplished through a proton pump located on the inner membrane of mitochondria, which can pump protons (H+) from the matrix into the intermembrane space. The transmembrane transport of protons accumulates a large amount of protons in the intermembrane space of mitochondria, forming a transmembrane potential across the inner membrane of mitochondria, known as the mitochondrial membrane potential. When protons return, they drive the production of ATP, the most important energy currency in living organisms. 95% of ATP in the human body is provided by mitochondria.
Under normal circumstances, the inner membrane potential of mitochondria is high and remains at a negative potential, while the outer membrane potential is low and remains at a positive potential. When certain factors cause obstacles in the electron transfer process of mitochondrial respiratory chain, affecting the formation of proton (H+) transmembrane gradient in the matrix, it will lead to a decrease in the mitochondrial membrane potential, i.e. depolarization, which is positive outside and negative inside. Numerous studies have shown that the decrease in mitochondrial membrane potential is related to autophagy, apoptosis, or necrosis. Mitochondrial membrane potential dysfunction, even subtle abnormal changes, may greatly affect the biological activity of cells and cause various diseases (Alzheimer's disease, diabetes, cancer, etc.).
Therefore, mitochondrial membrane potential is one of the important indicators for evaluating the normal function of mitochondria. It not only reflects the integrity of mitochondrial function, but also has important effects on the synthesis of TP, ion transport, regulation of cell apoptosis, and antioxidant activity. At the same time, it also reflects the health status of cells and has important significance for their survival and development. In addition, detecting changes in mitochondrial membrane potential is of great significance for biological research and medical diagnosis. So, we should pay attention to the changes in mitochondrial membrane potential and take corresponding measures to protect the normal function of mitochondria.
MMP Detection Methods
Currently, fluorescent probes represent the most common approach for evaluating mitochondrial function through membrane potential assessment. These probes—including JC-1, Rh123, TMRM, TMRE, and DiOC6—typically consist of lipophilic cationic compounds that accumulate in the mitochondrial membrane matrix space inversely proportional to ΔΨm.
JC-1 Probe
JC-1 is an ideal fluorescent probe widely used for detecting mitochondrial membrane potential in cells, tissues, or purified samples. Its unique property of forming aggregates (red fluorescence) in healthy mitochondria and monomers (green fluorescence) in depolarized mitochondria allows for both qualitative and quantitative assessment of MMP.
Rhodamine 123 Probe
Rh123 is a cell-permeant, cationic, fluorescent dye that is readily sequestered by active mitochondria without cytotoxic effects. Its fluorescence intensity decreases as the membrane potential decreases, making it useful for measuring MMP changes.
TMRM/TMRE Probes
Tetramethylrhodamine methyl ester (TMRM) and ethyl ester (TMRE) are cell-permeant dyes that distribute between mitochondria and the cytosol according to the Nernst equation. They are ideal for quantitative measurements of membrane potential.
DiOC6 Probe
DiOC6(3) is a carbocyanine dye that stains mitochondria in live cells and its accumulation is dependent upon the mitochondrial membrane potential. It's commonly used in flow cytometry applications.
JC-1 Detection Principle
JC-1 dye accumulates in mitochondria in a potential dependent manner, exhibiting multi-color fluorescence emission spectra and allowing for proportional semi quantitative evaluation of mitochondrial depolarization. In normal mitochondria, JC-1 aggregates in the mitochondrial matrix to form a polymer, which emits strong red fluorescence; When compounds induce mitochondrial membrane potential collapse, JC-1 can only exist in the cytoplasm as a monomer due to the decrease or loss of membrane potential, emitting green fluorescence. This means that in healthy cells, a small amount of green fluorescence can appear, with the majority being red fluorescence; Most cells with depolarization of mitochondria only display green fluorescence. Therefore, changes in mitochondrial membrane potential can be reflected by changes in fluorescence color, or the degree of mitochondrial depolarization can be measured by the ratio of red/green fluorescence intensity.
The excitation wavelength of JC-1 monomer is 488 or 514nm, emitting green fluorescence with a wavelength of around 529nm; The maximum excitation wavelength of JC-1 polymer is 585nm, emitting red fluorescence at a wavelength of around 590nm. JC-1 can be detected using fluorescence microscopy, laser confocal microscopy, fluorescence spectrophotometer, or flow cytometry.
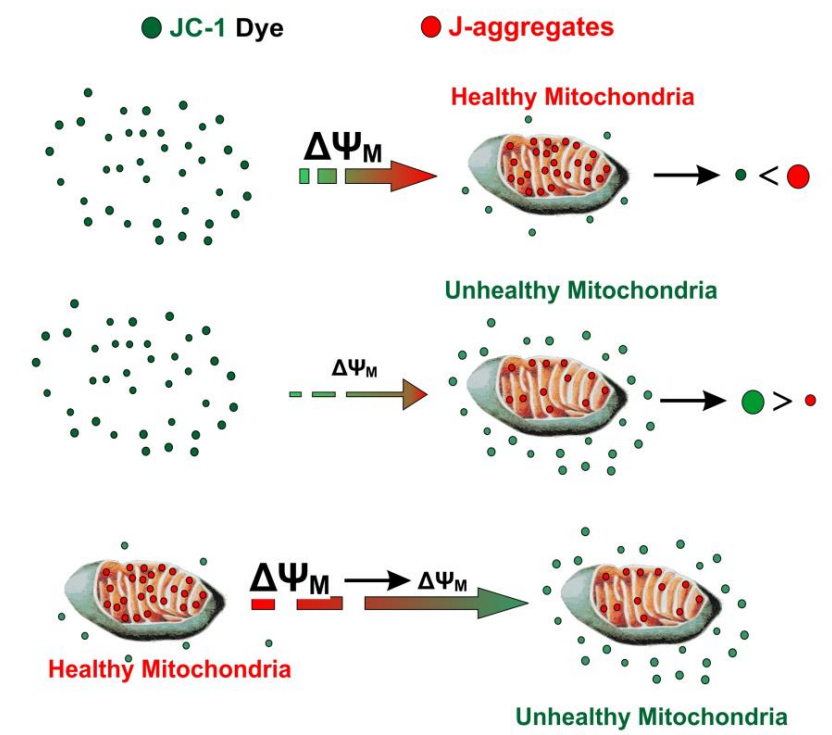
Figure 2: JC-1 Entry into Mitochondria and Aggregate Formation (Sivandzade et al., 2019)
Experimental Procedure
CCCP is a reversible proton gradient uncoupling agent that can rapidly reduce the electrochemical potential across the mitochondrial membrane, leading to rapid depolarization of intracellular mitochondria. Therefore, cells treated with CCCP can be used as positive controls for JC-1 detection. After CCCP treatment, the mitochondrial membrane potential will be completely lost, and JC-1 staining will show green fluorescence, while normal cells should show red fluorescence after JC-1 staining.
1. Cell staining
1.1 Suspended cells
(1) Cell plating: Taking a 6-well plate as an example, resuspend 1 million cells in 1mL of culture medium per well, and repeat the same process for other culture vessels. You can also choose the appropriate density for plating according to your own cell type.
(2) Set up positive control: Take an appropriate amount of CCCP to treat cells and incubate them in a cell culture incubator at 37 ℃ for 5 minutes.
(3) JC-1 staining: Add an appropriate amount of JC-1 and incubate in a cell culture incubator at 37 ℃ for 15 minutes. Generally, 15 minutes is sufficient for sufficient staining.
(4) Centrifuge: After incubation at 37 ℃, centrifuge 400g at 4 ℃ for 3-4 minutes, discard the supernatant, and be careful not to remove cell sediment as much as possible.
(5) Washing: Wash twice with 1 × PBS, centrifuge 400g at 4 ℃ for 3-4 minutes.
(6) Result detection: Resuspend the cells in 500 μ L PBS and observe them using a fluorescence microscope or laser confocal microscope, or analyze them using a fluorescence spectrophotometer or flow cytometer (green fluorescence: Ex/Im=510/527nm; Red fluorescence: Ex/Im=585/590nm).
1.2 Adherent cells
If using a fluorescence spectrophotometer or flow cytometer to detect adherent cells, the adherent cells can be digested and collected first, resuspended, and then the detection method for suspended cells can be referred to. If using a fluorescence microscope or laser confocal microscope for detection: simply refer to the steps and methods of the suspended cell experiment.
Precautions
CCCP is a mitochondrial electron transfer chain inhibitor that is harmful to the human body. During the experiment, it is necessary to wear experimental clothing and disposable gloves, and be careful with protection.
JC-1 is photosensitive, and all dyeing steps should avoid exposure to strong light during the operation process; Unused storage solution should be stored away from light and avoid repeated freezing and thawing.
After JC-1 staining is completed, subsequent result analysis should be carried out immediately to reduce various possible errors.
References
[1] Sivandzade F, Bhalerao A, Cucullo L. Analysis of the Mitochondrial Membrane Potential Using the Cationic JC-1 Dye as a Sensitive Fluorescent Probe. Bio Protoc. 2019 Jan 5; 9(1):e3128.
[2] Zorova LD, Popkov VA, Plotnikov EY, et al. Mitochondrial membrane potential. Anal Biochem. 2017 Jul 1; 552:50-59.
