JC-1
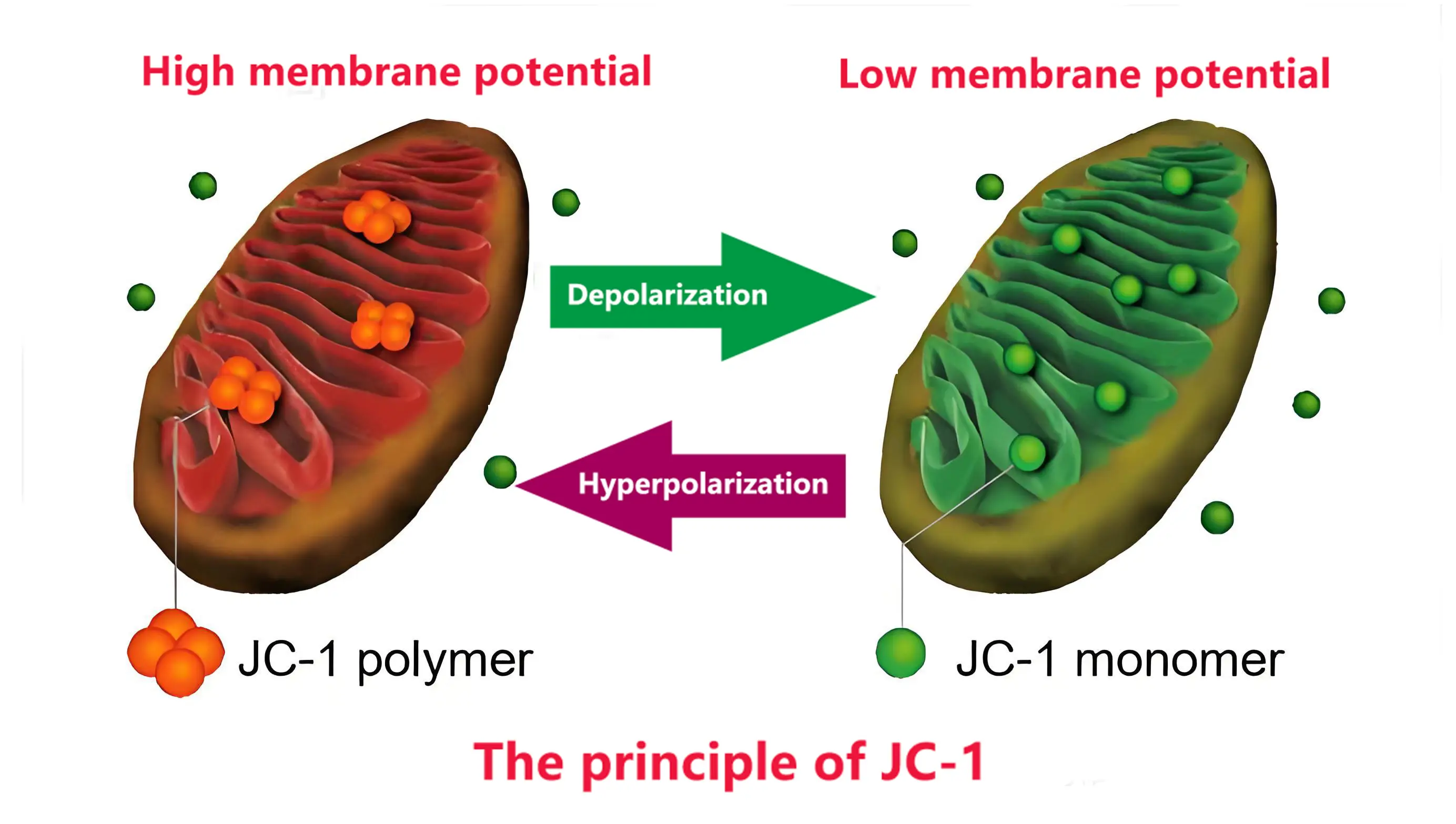
The principle of JC-1 detection
JC-1 (also known as CBIC2 (3)) is a fluorescent probe used to detect mitochondrial membrane potential (Δ Ψm). Its molecular formula is C25H27Cl4IN4, molecular weight is 652.23, CAS number is 3520-43-2, and purity is over 95%. This substance is widely used for membrane potential detection in cells, tissues, or purified mitochondria.
JC-1 reflects the mitochondrial membrane potential state through fluorescence color changes: at high potential, it aggregates in the mitochondrial matrix to form J-aggregates that emit red fluorescence (excitation/emission wavelength 585/590nm), and at low potential, it exists in monomeric form and emits green fluorescence (excitation/emission wavelength 514/529nm). The ratio of red to green fluorescence intensity can be used as an indicator of mitochondrial depolarization, and is commonly used to detect fluorescence changes caused by membrane potential decline in the early stage of cell apoptosis. This characteristic makes it an important tool for studying mitochondrial function and cell apoptosis mechanisms.
The decrease in mitochondrial membrane potential is a hallmark event in the early stage of cell apoptosis. The transition of JC-1 from red fluorescence to green fluorescence can easily detect the decrease in cell membrane potential, and the transition of JC-1 from red fluorescence to green fluorescence can also be used as an early detection indicator of cell apoptosis.
JC-1 mitochondrial membrane potential detection protocol
Preparation before the experiment
1. Materials and reagents
- JC-1 probe, culture medium, reagent kit, ultrapure water
- JC-1 staining buffer, PBS buffer, etc.
2. Equipment
- Fluorescence microscope (equipped with 488 nm excitation light, capable of detecting 530 nm and 590 nm fluorescence), flow cytometer (set 488 nm excitation light, FL-1 channel detects 530 nm green fluorescence, FL-2 channel detects 590 nm red fluorescence), temperature control equipment (37 ℃ incubator), pipette
3. Cell preparation
Ensure that cells are in logarithmic growth phase to achieve optimal membrane potential state.
Experimental steps
1. Preparation of JC-1 staining working solution
According to the instructions of the kit, dissolve the JC-1 probe in DMSO and dilute it with serum-free medium to the desired concentration (usually 2.5-10 µ g/mL).
Each well of the six well plate requires 1 mL of staining working solution, and the cell suspension requires 0.5 mL of staining working solution for every 500000 to 1 million cells.
Ensure that JC-1 is fully dissolved and mixed.
2. Cell staining
Add an appropriate amount of JC-1 working solution to the cell culture medium and mix gently.
Incubate the cell plate in a 37 ℃, 5% CO2 incubator for 15-30 minutes (adjusted according to cell type).
After staining, wash the cells 2-3 times with pre cooled PBS to remove unbound probes.
3. Cell processing
For adherent cells, collect the cell suspension after mild treatment with trypsin.
For suspended cells, resuspend, incubate, centrifuge, and wash according to the steps.
Finally, resuspend the cells in JC-1 staining buffer and prepare for detection.
4. Detection and Data Collection
Fluorescence microscopy detection: Observe cells with excitation wavelength of 488 nm, red fluorescence emission wavelength of 590 nm, and green fluorescence emission wavelength of 530 nm. Healthy cells exhibit red fluorescence, while damaged cells exhibit green fluorescence.
Flow cytometry detection: Measure the red green fluorescence ratio of the cell population, calculate the red/green fluorescence ratio to evaluate changes in mitochondrial membrane potential.
Result analysis
Generate a red/green fluorescence ratio chart through software analysis.
When detected by flow cytometry, FL-1+and FL-2+are normal cells, while FL-1+and FL-2- are apoptotic cells.
Precautions
1. Dissolution and preservation of JC-1:
JC-1 solidifies at low temperatures and needs to be dissolved in a water bath at 20-25 ℃ before use.
After staining is completed, try to complete the test within 30 minutes, and unused staining solution should be stored in an ice bath.
2. Operating conditions:
Maintain 37 ℃ during the dyeing process to avoid temperature fluctuations.
During washing, maintain JC-1 staining buffer at around 4 ℃ to achieve better washing results.
3. Experimental repeatability and reliability:
• Avoid the influence of external factors (such as pH changes and temperature fluctuations) on cells.
Ensure pH consistency of JC-1 staining buffer.
4. Safety and compensation adjustment:
When using CCCP (mitochondrial electron transport chain inhibitor), precautions should be taken.
When using a flow cytometer for detection, pay attention to compensation adjustment and use compensation microspheres or FITC/PE dye antibodies for calibration.
FAQ for JC-1 detection
Sure, but it is not possible to directly detect the tissue. The tissue can be prepared as a single-cell suspension first, and then tested according to the steps of suspending cells. In addition, it is important to note that the process of preparing single-cell suspensions can easily cause damage to cells, and the preparation plan needs to be explored to avoid false positives caused by changes in mitochondrial membrane potential due to cell digestion; Mitochondria can also be extracted from tissues, incubated with JC-1, and the experimental results can be detected using a fluorescence enzyme-linked immunosorbent assay (ELISA) reader.
Not recommended. Contact between adherent cells may result in uneven contact between cells and dyes, which can affect the entry of JC-1 dye into cells, especially when cell growth is dense. Recommend digesting cells before incubating JC-1.
No, the sample for JC-1 experiment must be live cells.
No, JC-1 is used for the detection of live cell samples and cell death occurs after fixation; In addition, leaving it for too long after staining can cause fluorescence quenching. It is recommended to complete the detection within 30 minutes of staining.
