Human TREM2 Activity Antibody
Alzheimer's Disease (AD) is one of the most common neurodegenerative diseases worldwide. Clinically, it is characterized by the presence of extracellular amyloid plaques and intracellular neurofibrillary tangles, resulting in neuronal dysfunction and cell death. Triggering receptor expressed on myeloid cells 2 (TREM2), a protein localized at the membrane of innate immune cells, including microglia in the brain, has been genetically linked to AD, with specific variants increasing disease risk by as much as threefold (1,2). The TREM2 receptor is a single-pass type I membrane glycoprotein that consists of an extracellular immunoglobulin-like domain, a transmembrane domain, and a cytoplasmic tail. Upon activation, TREM2 interacts with the tyrosine kinase-binding protein DNAX-activating protein 12 (DAP12, TYROBP) to form a receptor-signaling complex. The DAP12 protein structure consists of a short extracellular domain, a transmembrane domain, and a cytoplasmic immunoreceptor tyrosine-based activation motif (ITAM) (2-9). ITAMs function as a binding site for tyrosine kinases, including spleen tyrosine kinase (Syk). Syk is comprised of two tandem amino-terminal Src homology (SH) 2 domains separated by an SH2-kinase linker, and a C-terminal tyrosine kinase domain, separated from the SH2 domains by an inter-domain linker. When Syk binds to an ITAM, it changes conformation, allowing for residues within the inter-domain linker region, including Tyr352, to become phosphorylated. Residues within the activation loop subsequently become phosphorylated, leading to full Syk activation. Tyr525 and Tyr526 are located in the activation loop of the Syk kinase domain and phosphorylation at these residues (equivalent to Tyr519/520 of mouse Syk) is essential for Syk function (10-12). This activation can lead to the mediation of a variety of cellular responses, including proliferation, differentiation, inflammation, and phagocytosis. Evidence suggests that TREM2 and DAP12 may act in a Syk-dependent manner to promote microglial survival, proliferation, and phagocytosis, which may be important in the context of AD pathology (13).
There is also evidence that these processes may be regulated via crosstalk between TREM2 and the cell surface receptor CD33, a sialic acid-binding Ig-like lectin (Siglec-3) type I transmembrane protein. Much like TREM2, CD33 has been identified as a risk gene in AD. CD33 binds preferentially to alpha-2, 6-linked sialic acid, which can be found in sialylated gangliosides in the brain. Activation of CD33 has been shown to be inhibitory to a variety of cellular processes. Evidence suggests that TREM2 may act downstream of CD33 and that TREM2-dependent microglial signaling in AD may be directly inhibited by CD33 activation (14-17).
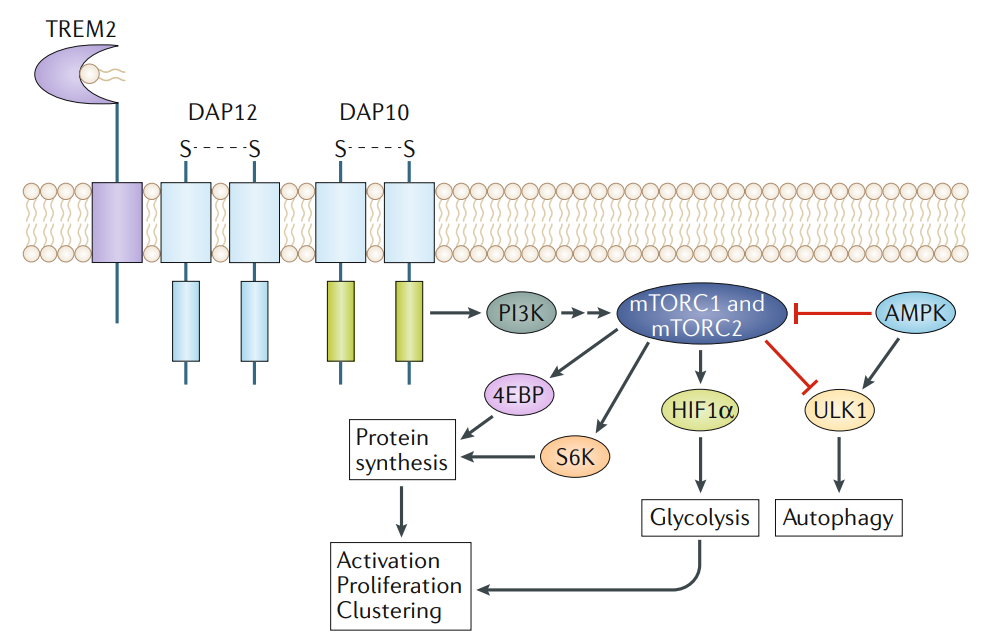
Signalling pathways linking TREM2 activation and energetic cellularmetabolism
Relevant Antibodies
| Catalog# | Product Name | Application | Reactivity |
|---|---|---|---|
| APRab03407 | TREM2 Rabbit Polyclonal Antibody | WB,IHC-P,ICC/IF | Human,Mouse,Rat |
| AMRe08370 | CD33 (18N8) Rabbit Monoclonal Antibody | WB,IHC-P,ICC/IF,IP,IF-P | Human |
| AMRe86818 | Syk (DGR14958) Rabbit mAb | WB, IHC-P, ICC/IF | Human,Mouse |
| AMRe09784 | DAP12 (15G16) Rabbit Monoclonal Antibody | WB,ICC/IF,FC | Human |
| APS0635 | HRP-conjugated Polyclonal Goat Anti-Rabbit IgG(H+L) Secondary Antibody | ELISA,WB,Dotblot | Mouse |
| AMRe80004 | GAPDH (12R9) Rabbit Monoclonal Antibody | WB,ELISA | Human,Mouse,Rat,Rabbit,Dog,Monkey |
Related Products
Super-sensitive ECL chemiluminescent reagent
References
- Nguyen, A.T. et al. (2020) Acta Neuropathol 140, 477-493.
- Gratuze, M. et al. (2018) Mol Neurodegener 13, 66.
- Jonsson, T. et al. (2013) N Engl J Med 368, 107-16.
- Jay, T.R. et al. (2017) Mol Neurodegener 12, 56.
- McQuade, A. et al. (2020) Nat Commun 11, 5370.
- Schlepckow, K. et al. (2020) EMBO Mol Med 12, e11227.
- Zhao, Y. et al. (2018) Neuron 97, 1023-1031.e7.
- Colonna, M. (2003) Nat Rev Immunol 3, 445-53.
- Lanier, L.L. et al. (1998) Nature 391, 703-7.
- Zhang, J. et al. (2000) J Biol Chem 275, 35442-7.
- Mansueto, M.S. et al. (2019) J Biol Chem 294, 7658-7668.
- Grädler, U. et al. (2013) J Mol Biol 425, 309-33.
- Turner, M. et al. (2000) Immunol Today 21, 148-54.
- Karch, C.M. et al. (2012) PLoS One 7, e50976.
- Griciuc, A. et al. (2013) Neuron 78, 631-43.
- Griciuc, A. et al. (2019) Neuron 103, 820-835.e7.
- Salminen, A. et al. (2021) Neurochem Int 150, 105186.
- Ulland TK, Colonna M. (2018). 14(11):667-675.
Voisey
Voisey is a technical support specialist at EnkiLife, proficient in immunology and cell biology. She is committed to providing customers with professional and efficient technical support. Additionally, she is involved in research on customers' fields of study and designs highly cost-effective solutions for them.
