Persistent Activation of TRPM4 Triggers Necrotic Cell Death Characterized by Sodium Overload

Published in Nature Chemical Biology, this study represents a major breakthrough in the field of cell death. It is the first to identify a novel form of programmed necrosis characterized by sodium overload — NECSO(Necrosis by Sodium Overload). The research defines transient receptor potential cation channel TRPM4 as the core regulator of this process, elucidates its molecular mechanism, species specificity and disease association, and identifies the first inhibitor of NECSO, providing a novel target and strategy for cell death regulation and the treatment of related diseases.
1. Research Background
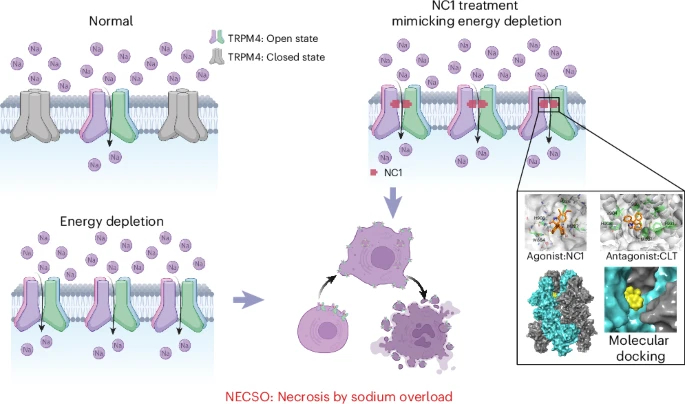
Excessive sodium ions are closely associated with many human diseases (e.g., hypertension, cardiovascular diseases, cerebral edema). While overloads of calcium, iron, copper and other ions have been confirmed to directly cause cell death, it remains unclear whether sodium overload has a similar direct pathogenic mechanism. This study identified a chiral small‑molecule compound named “Necrocide 1” (NC1), which induces a unique immunogenic necrotic cell death that is not blocked by known inhibitors of apoptosis, necroptosis, autophagy or ferroptosis.
Using genome‑wide CRISPR‑Cas9 screening, the research team discovered that transient receptor potential cation channel subfamily M member 4 (TRPM4) is the key target of NC1. NC1 triggers massive sodium influx by specifically activating human TRPM4 (hTRPM4), thereby inducing cell necrosis. This novel cell death modality is termed “sodium overload‑induced necrosis (NECSO)”.
2. Research Results
2.1 TRPM4 is a Key Factor in NC1‑Induced Cell Death
Genome‑wide CRISPR‑Cas9 screening revealed that the TRPM4 gene was the most highly enriched candidate gene in NC1‑resistant cells. Knockout of TRPM4 in MCF7 cells completely blocked NC1‑induced cell death and lactate dehydrogenase (LDH) release, whereas re‑expression of human TRPM4 restored sensitivity to NC1. In addition, the ion channel activity of TRPM4 is essential for NC1‑induced necrosis, as re‑expression of the ion‑impermeable mutant D984A failed to rescue the necrotic phenotype.
Experiments confirmed that NC1 binds directly to TRPM4. Cellular thermal shift assay (CETSA) showed that NC1 stabilizes the TRPM4 protein at high temperatures, and surface plasmon resonance (SPR) analysis measured a binding affinity of 0.88 μM. Whole‑cell patch‑clamp recordings showed that NC1 elicits TRPM4‑dependent currents with persistent activation that persists even after washout, indicating that NC1 induces persistent and irreversible activation of the TRPM4 channel.
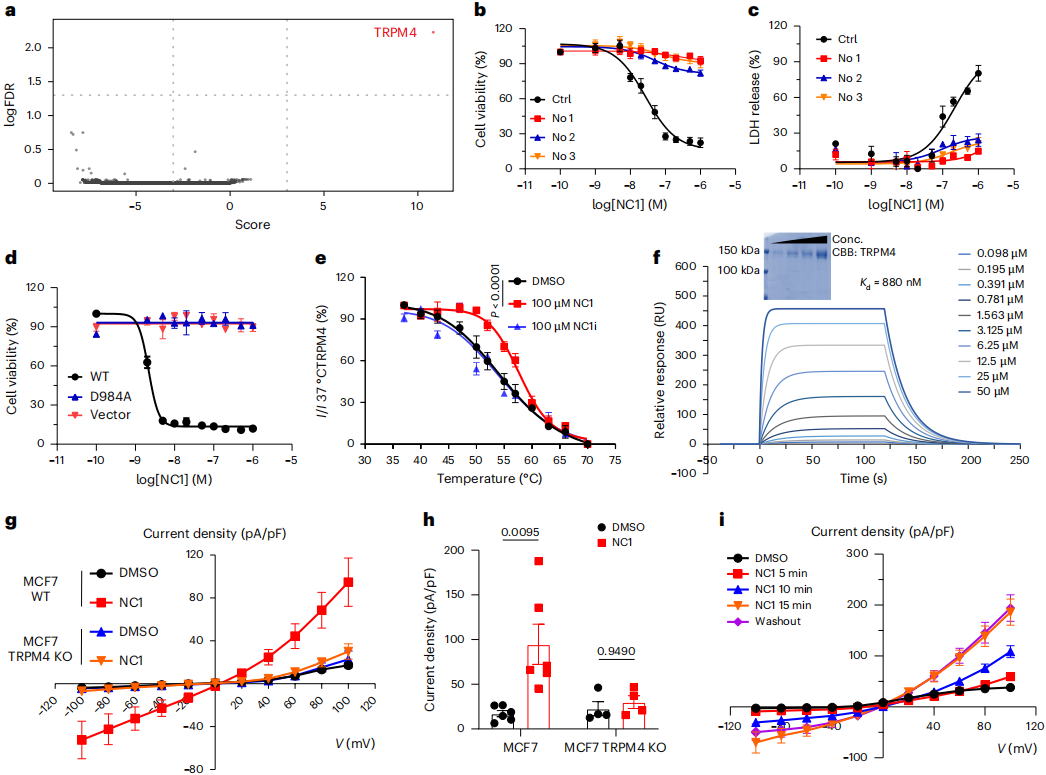
Figure 1 | NC1 triggers a unique cell death pathway dependent on TRPM4.
2.2 NC1‑Induced Necrosis via TRPM4 Depends on Sodium Influx
TRPM4, a non‑selective monovalent cation channel, induces massive sodium influx upon activation. Inductively coupled plasma mass spectrometry (ICP‑MS) analysis showed a significant increase in intracellular sodium levels after NC1 treatment. Substitution of sodium ions in the extracellular medium revealed that sodium depletion markedly blocked cell death, whereas inhibition of potassium efflux had no significant effect, confirming that sodium influx rather than potassium efflux is the key factor leading to necrosis. This sodium overload‑driven cell death is defined as “sodium overload‑induced necrosis (NECSO)”.
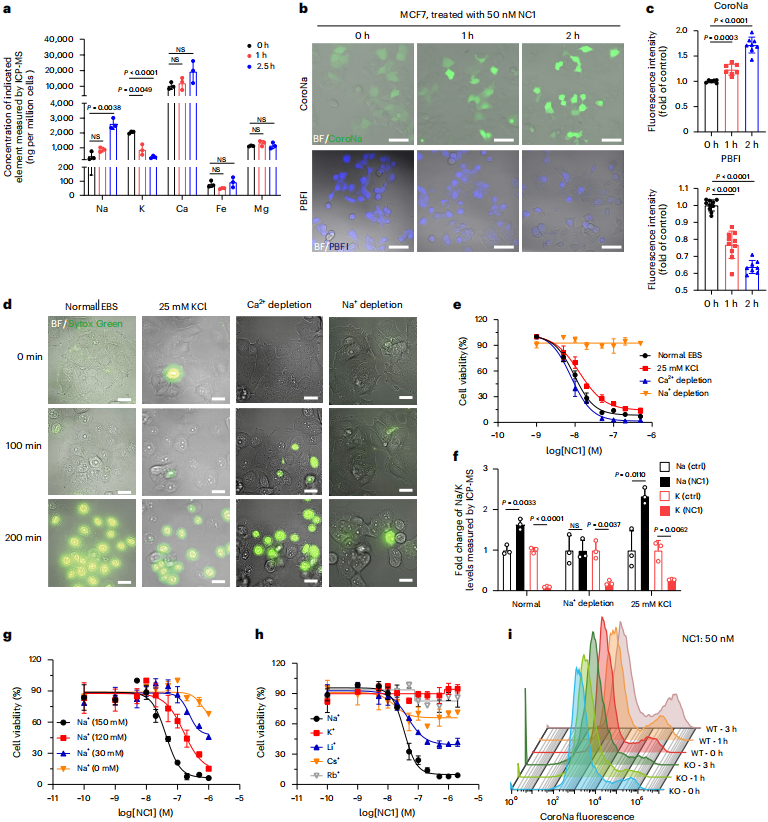
Figure 2 | Sodium influx is critical for NC1‑induced cell death.
2.3 NC1 Specifically Targets Human TRPM4
The study shows that NC1 specifically activates human TRPM4 but not mouse TRPM4 (mTRPM4), despite 88.4% sequence similarity. By constructing human‑mouse chimeric proteins, the researchers identified the transmembrane region as the key domain determining NC1 sensitivity. These results indicate that NC1 is a highly specific agonist of human TRPM4 and induces sodium overload necrosis only in cells expressing hTRPM4.
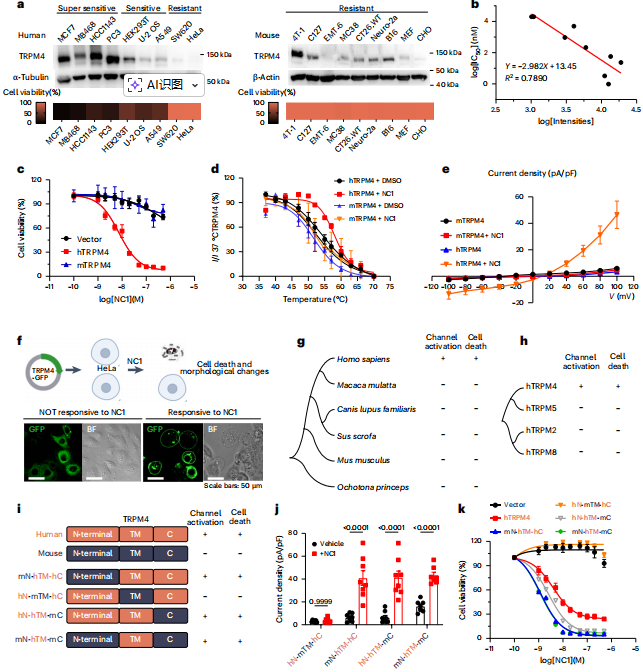
Figure 3 | NC1 specifically targets human TRPM4 through interaction with the transmembrane domain
2.4 Molecular Docking Identifies Key Amino Acid Residues for NC1‑Mediated TRPM4 Activation
Molecular docking and mutagenesis analyses revealed that NC1 binds to a vanilloid‑binding pocket (VBP)‑like site in the transmembrane domain of TRPM4, involving key residues on the S3, S4, S5 and S6 helices. Mutation of key residues (e.g., W864, H908, F936) to alanine disrupts NC1 binding and channel activation, confirming that this binding pocket is an essential structure for NC1‑induced TRPM4 activation.
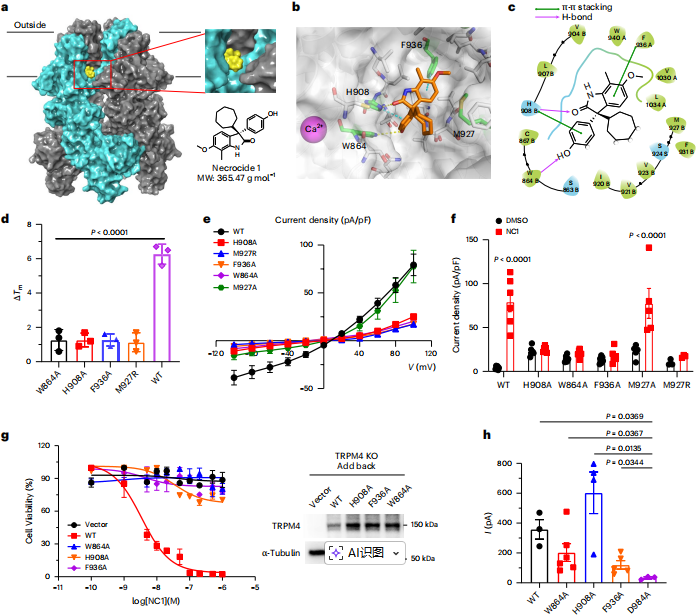
Figure 4 | Molecular docking and mutagenesis reveal key residues in TRPM4 essential for NC1 activation.
2.5 Energy Depletion‑Induced Necrosis Shares Common Features with Sodium Overload Necrosis
The study found that energy depletion (e.g., 2DG + NaN₃ treatment) also activates TRPM4 and triggers sodium influx and necrosis, which share similar genetic and chemical intervention characteristics with NC1‑induced NECSO. In addition, TRPM4 gain‑of‑function mutants associated with heart disease show higher sensitivity to cell death induced by NC1 or energy depletion.
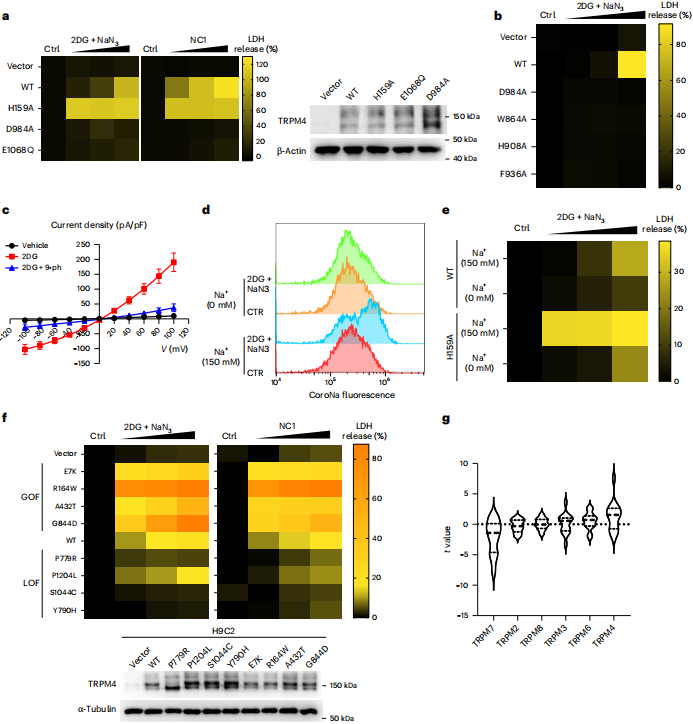
Figure 5 | Energy depletion‑induced cell death shares common features with sodium overload necrosis
2.6 Identification of Potential Inhibitors of NECSO
The study identified clotrimazole (CLT) and dihydropyridine compounds (DHPs) as inhibitors of NECSO. CLT inhibits NECSO by direct competitive binding to TRPM4, while DHPs act indirectly as L‑type voltage‑gated calcium channel blockers. These inhibitors effectively protect cardiomyocytes from cell damage associated with TRPM4 gain‑of‑function mutations.
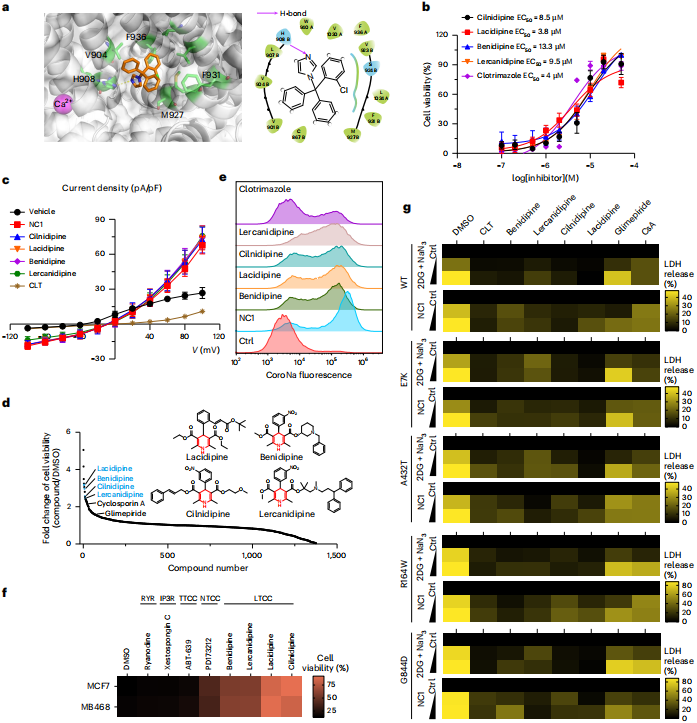
Figure 6 | DHPs and CLT function as small‑molecule inhibitors of NECSO
3. Summary
This study not only defines a novel form of necrotic cell death driven by sodium overload, NECSO, but also uncovers its core executor TRPM4 and the mechanism of its specific agonist NC1. This discovery links cell death research to sodium homeostasis, providing new insights into the pathogenesis of ischemic injury, heart failure and other TRPM4‑related diseases. Furthermore, CLT and DHPs as potential therapeutic agents demonstrate the feasibility of intervening in cell death by targeting TRPM4 or its related pathways.
Literature Source
Fu W, et al. Persistent activation of TRPM4 triggers necrotic cell death characterized by sodium overload. Nat Chem Biol. 2025 Aug;21(8):1238‑1249. doi: 10.1038/s41589‑025‑01841‑3. Epub 2025 Feb 6. PMID: 39915626.
Antibodies Involved in This Study Are Available from EnkiLife:
| Target | Catalog# | Product Name | Reactivity | Application |
|---|---|---|---|---|
TRPM4 | APRab19320 | TRPM4 Rabbit Polyclonal Antibody | Human,Mouse,Rat | WB |
Caspase‑9 | AMRe07987 | Caspase‑9 (13Z10) Rabbit Monoclonal Antibody | Human,Mouse | WB,IHC‑P,ICC/IF,FC,IP,IF‑P |
α‑tubulin | AMM80017 | α‑tubulin Monoclonal Antibody(8F11) | Human,Mouse,Rat | WB,IHC‑P,IF‑P,IF‑F,IF‑ICC,IP,ELISA |
α‑tubulin | APRab80018 | α‑tubulin Polyclonal Antibody | Human,Mouse,Rat | WB,ELISA |
β‑actin | AMRe80020 | β‑actin Rabbit Monoclonal Antibody | Human,Mouse,Rat | WB,ELISA |
β‑Tubulin | AMM80021 | β‑Tubulin Monoclonal Antibody(5G3) | Human,Rat,Mouse,Mk,Dg,Ch,Hamster,Rb,sheep,Insect,Yeast | WB,IF,IHC‑p,ELISA |
Related Products

