Literature Sharing: The Integrated Stress Response Mediates Tumor Immune Evasion via LCN2
1. Research Background
Tumor immune evasion is the core bottleneck restricting the efficacy of cancer immunotherapy and a current research hotspot and challenge in the field of tumor immunology. In recent years, the advent of immunotherapeutic approaches such as immune checkpoint inhibitors (ICIs) has brought new therapeutic hope to patients with various solid tumors. However, clinical data show that more than 50% of patients still do not respond to immunotherapy or develop resistance. The fundamental reason lies in tumor cells evading recognition and elimination by the immune system through multiple molecular mechanisms. The Integrated Stress Response (ISR), as a core regulatory pathway for cells to cope with microenvironmental stressors such as endoplasmic reticulum stress and metabolic pressure, plays a key role in tumor cell survival, proliferation, and immune evasion. Among them, Activating Transcription Factor 4 (ATF4), as the most critical transcription factor downstream of ISR, can participate in various biological processes such as tumor cell metabolic reprogramming, anti-apoptosis, and immune suppression by regulating the expression of downstream target genes. Existing studies have confirmed that ATF4 is highly expressed in various malignant solid tumors such as lung cancer, pancreatic cancer, and melanoma, and its expression level is closely related to tumor malignancy, patient prognosis, and immunotherapy resistance. However, the specific molecular mechanisms by which ATF4 regulates tumor immune evasion, key downstream effector molecules, and their targets have not been fully elucidated, which has become an important bottleneck for the clinical translation of basic research in this field. The study "The integrated stress response promotes immune evasion through lipocalin 2" published by scholars such as Bossowski J, Papagiannakopoulos T, and Koide S in the top international journal Nature conducted a systematic and in-depth investigation on this urgent scientific problem. Focusing on clinically common solid tumors with high immunotherapy resistance rates, such as lung cancer, pancreatic cancer, and melanoma, and combining in vitro and in vivo experiments, clinical sample analysis, and big data mining, this study for the first time revealed the molecular mechanism by which Lipocalin 2 (LCN2), as a key effector molecule downstream of the integrated stress response, mediates tumor immune evasion, and further confirmed the therapeutic potential of targeting LCN2.
2. Research Methods
To systematically clarify the molecular mechanism by which the integrated stress response mediates tumor immune evasion through LCN2, and to determine the clinical significance and therapeutic potential of targeting LCN2, this study constructed a multi-level research system by comprehensively applying various research methods. The study focused on KP mouse lung cancer cells, B16F10 melanoma cells, and KPC7 pancreatic cancer cells as core objects, using CRISPR-Cas9 technology to construct LCN2 and ATF4 knockout and overexpression cell lines, combined with custom ATF4 target gene CRISPR-Cas9 library in vivo screening, ChIP-qPCR, Western blot, and ELISA techniques to verify the integrated stress response-ATF4-LCN2 regulatory axis and the secretory properties of LCN2. For animal experiments, immunocompetent C57BL/6J mice and immunodeficient NSG mice were used to construct tumor transplantation models, combined with KPC genetically engineered mice and doxycycline-inducible shRNA silencing system to explore the role of LCN2 in different immune environments and different tumor stages. Tumor microenvironment analysis adopted ExCITE-seq single-cell sequencing, flow cytometry, immunofluorescence, and other techniques to deeply analyze the changes in immune cell composition and function after LCN2 silencing.
3. Results Analysis
ATF4 Drives Tumor Progression by Inhibiting Anti-tumor Immunity
The study first verified the core role of ATF4 in tumor progression. ATF4-knockout KP lung cancer cells showed no significant difference in in vitro proliferation ability compared to wild-type cells, but in immunocompetent C57BL/6J mice, their tumor growth rate was significantly slowed, and mouse survival was significantly prolonged. In immunodeficient NSG mice, ATF4 knockout had no significant effect on tumor growth, suggesting that the tumor-promoting effect of ATF4 depends on the immune system. To further verify this conclusion, the study used the ISR inhibitor ISRIB (which can specifically antagonize phosphorylated eIF2α, thereby inhibiting ATF4 activity) to treat tumor-bearing mice. The results showed that ISRIB treatment could significantly inhibit tumor growth and prolong survival in immunocompetent mice, but could not work in NSG mice. These results together clearly outline the action logic of ATF4—it does not directly promote tumor cell proliferation, but rather by weakening the body's anti-tumor immune response, it paves the way for tumor growth, and also lays the foundation for subsequent exploration of downstream molecular targets of ATF4 that regulate immune evasion.
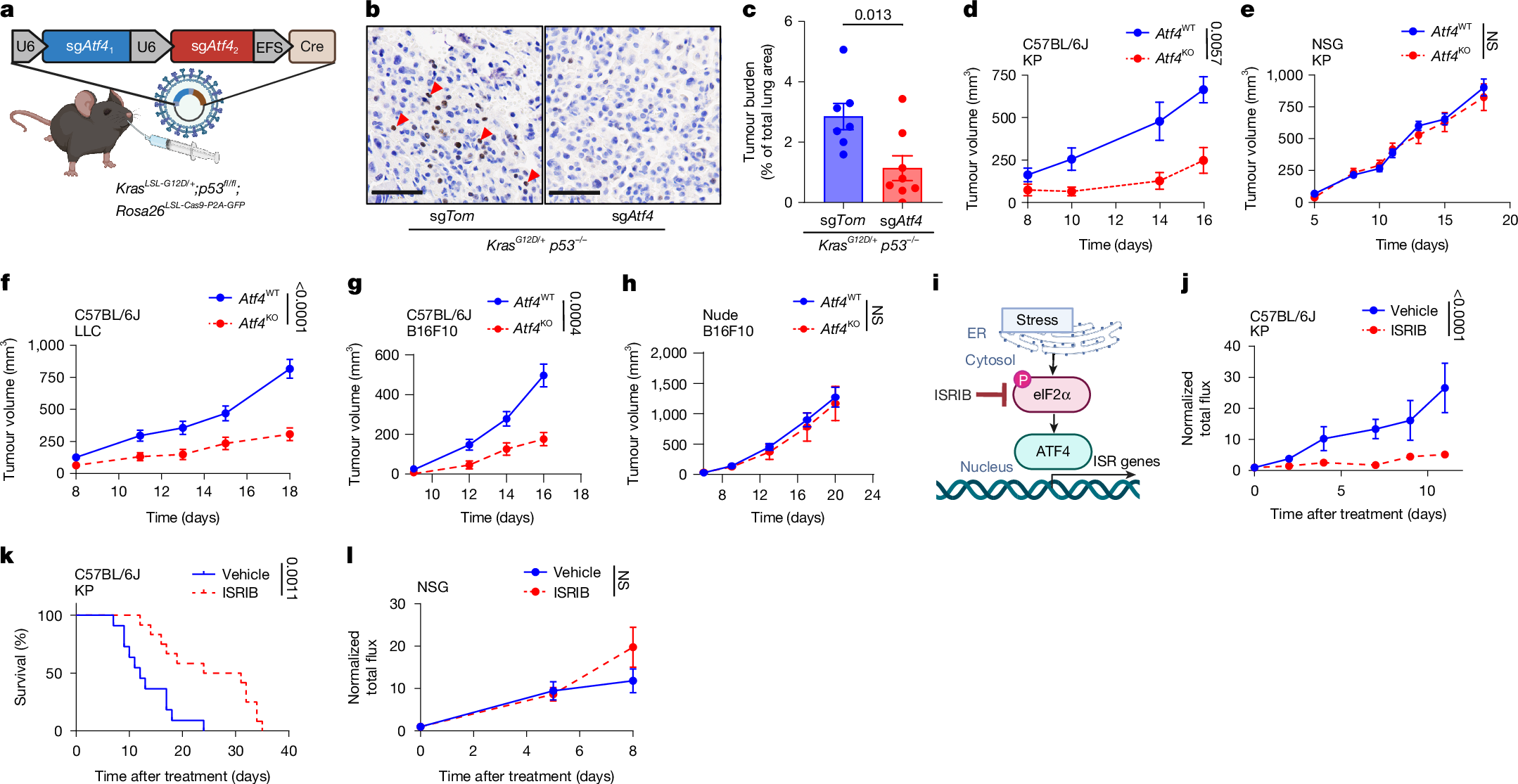
LCN2 is a Key Downstream Target of ATF4-Mediated Immune Evasion
To find the key factors driving immune suppression by ATF4, the study used a custom CRISPR-Cas9 library (containing 477 potential ATF4 target genes) to conduct negative screening in C57BL/6J mice, and found that LCN2 was one of the significantly depleted genes besides ATF4 itself, suggesting that LCN2 may be involved in ATF4-mediated immune evasion. Subsequent experiments further verified this hypothesis. Consistent with the phenotype of ATF4 knockout, LCN2-knockout KP cells showed significantly inhibited tumor growth in C57BL/6J mice, but no significant difference in NSG mice. Reintroducing LCN2 into LCN2-knockout cells restored tumor growth ability, and overexpressing LCN2 in ATF4-knockout cells could also rescue their tumor growth defects, directly proving that LCN2 is a key effector molecule downstream of ATF4, and the immune suppression effect of ATF4 depends on LCN2.
To further explore the key characteristics of LCN2's function, the study constructed multiple LCN2 mutants: mutants lacking the secretory signal peptide could not rescue the tumor growth defects of LCN2-knockout cells, while LCN2 mutants with lost iron-binding ability could function normally, indicating that the secretory function of LCN2 is key to its immune suppression effect, and is unrelated to its iron-binding ability. In addition, in the KPC genetically engineered mouse model, knockout of LCN2 via CRISPR-Cas9 significantly inhibited tumorigenesis. In other solid tumor models such as melanoma and pancreatic cancer, LCN2 knockout also inhibited tumor growth, indicating that the immune suppression effect of LCN2 is universal across tumor types. More importantly, using doxycycline-induced shRNA to silence LCN2 in established tumors could significantly slow tumor growth and prolong mouse survival, suggesting that LCN2 is a potential therapeutic target for advanced tumors.
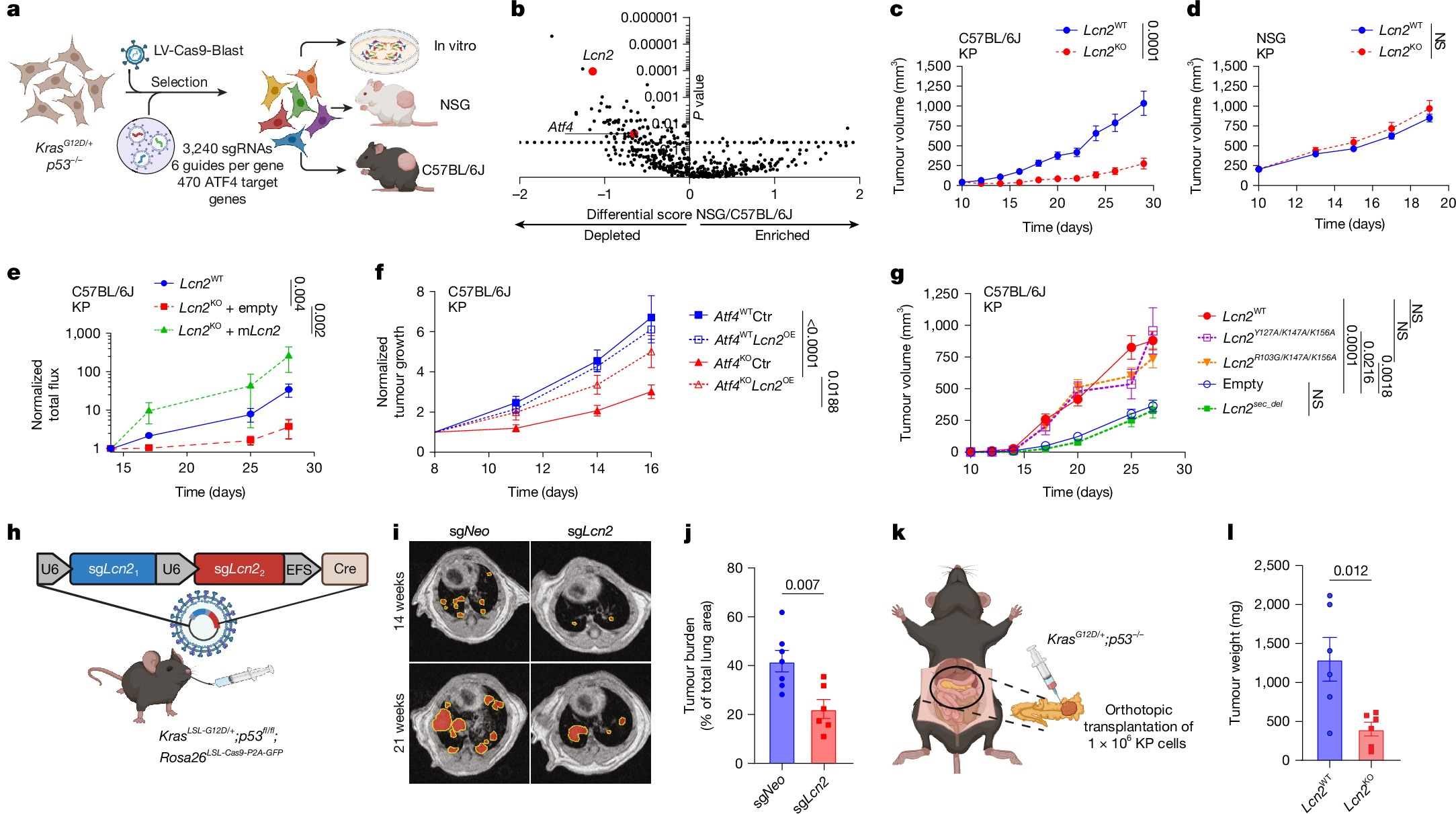
The ISR-ATF4 Axis Directly Regulates LCN2 Expression
The study further clarified the molecular mechanism by which ATF4 regulates LCN2 expression. Classical ISR inducers could significantly upregulate ATF4 protein levels and LCN2 expression, but this induction effect completely disappeared in ATF4-knockout cells. ISRIB treatment could block the upregulation of LCN2 by ISR inducers, but did not affect NF-κB pathway-mediated LCN2 expression, indicating that LCN2 expression is specifically regulated by the ISR-ATF4 axis. Clinically relevant metabolic inhibitors, such as the glutaminase inhibitor CB-839 and the mitochondrial respiration inhibitor phenformin, could also induce LCN2 expression, and this induction was dependent on ATF4. In vivo experiments showed that phenformin treatment could increase LCN2 expression in tumor cells of tumor-bearing mice, and was positively correlated with the proportion of ATF4-positive cells, while ISRIB treatment would reduce the proportion of ATF4-positive cells.
ChIP-qPCR experiments confirmed that ATF4 could directly bind to the promoter region of LCN2, similar to known ATF4 target genes Chac1 and Asns, further verifying the direct transcriptional regulatory effect of ATF4 on LCN2. TCGA database analysis showed that in lung cancer, pancreatic cancer, and melanoma, LCN2 expression was highly correlated with ATF4 transcriptional characteristics. In non-small cell lung cancer, LCN2 expression was also positively correlated with NF-κB characteristics, suggesting that LCN2 expression is co-regulated by both ATF4 and NF-κB pathways, but ATF4-mediated regulation plays a dominant role in tumor immune evasion.
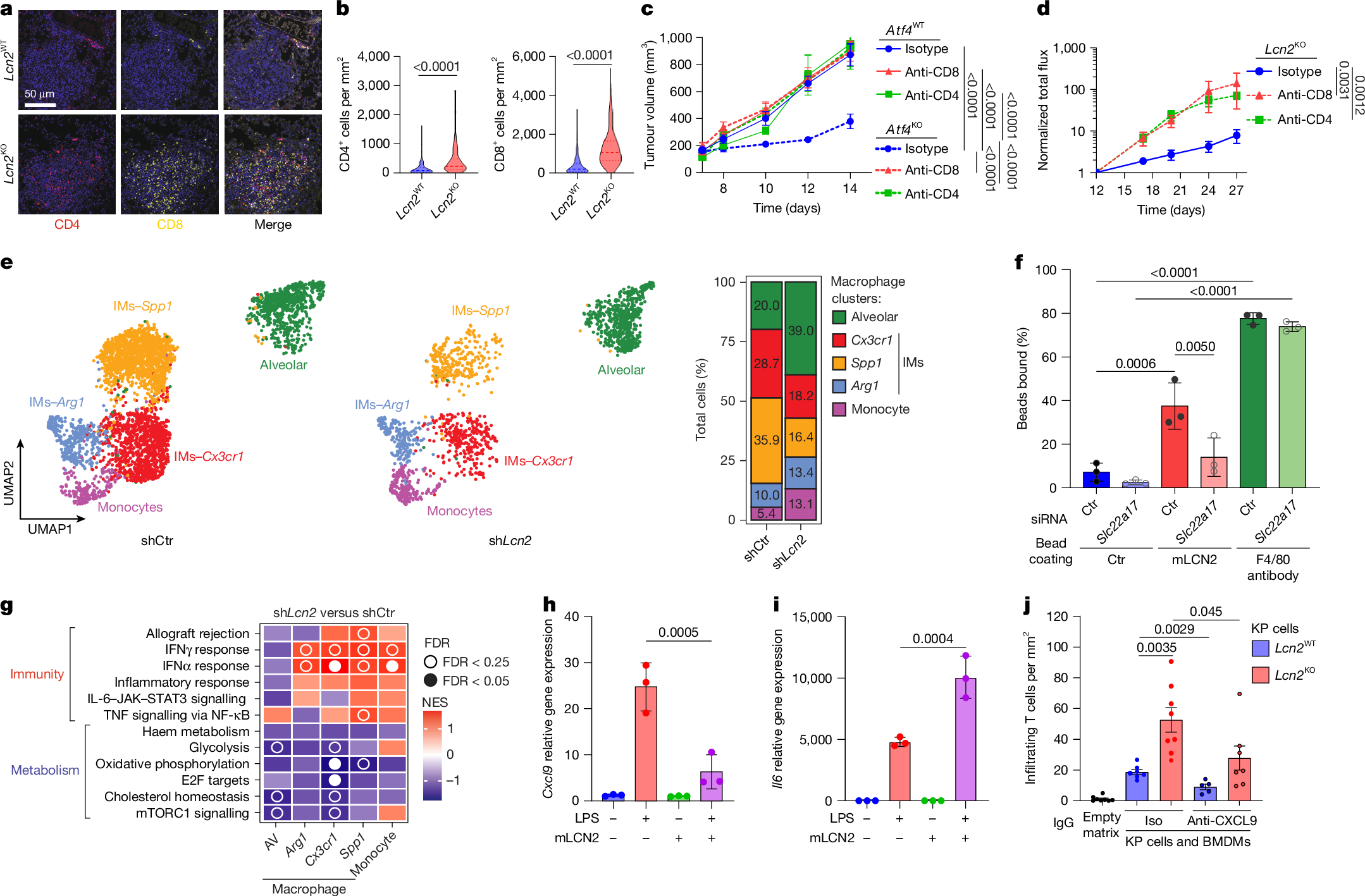
LCN2 Inhibits T Cell-Mediated Anti-tumor Immunity by Regulating Macrophage Phenotype
Since LCN2 is a secreted protein, its immune suppression effect must be achieved by regulating immune cells in the tumor microenvironment. The study found that in LCN2-knockout lung cancer tumors, the number of CD4+ and CD8+ tumor-infiltrating lymphocytes (TILs) increased significantly, while the number of CD4+FOXP3+ regulatory T cells (Treg) with immune suppression function decreased significantly. ISRIB treatment could also mimic this phenotype, increasing CD8+ TIL infiltration. More importantly, after depleting CD4+ or CD8+ T cells, the tumor growth inhibition effect brought by ATF4 knockout and LCN2 knockout completely disappeared, indicating that T cells are the core target of LCN2-mediated immune evasion.
ExCITE-seq single-cell analysis further revealed the regulatory details of LCN2 on immune cells in the tumor microenvironment. After LCN2 silencing, the proportion of macrophages and neutrophils in tumors decreased, the proportion of alveolar macrophages (AMs) increased, while the proportion of interstitial macrophage (IMs) subsets decreased. Further studies found that the receptor of LCN2, SLC22A17, is mainly expressed in macrophages, and LUMICKS AFS experiments confirmed that LCN2 can interact with macrophages by binding to SLC22A17. Functional analysis showed that LCN2 can inhibit the expression of the anti-tumor factor CXCL9 in macrophages and promote the expression of the pro-tumor factor IL-6, thereby inhibiting T cell infiltration, and knockout of SLC22A17 can block the regulatory effect of LCN2 on macrophages. 3D co-culture experiments further verified that LCN2-knockout cells can promote T cell infiltration and activation, while addition of recombinant LCN2 protein can reverse this effect.
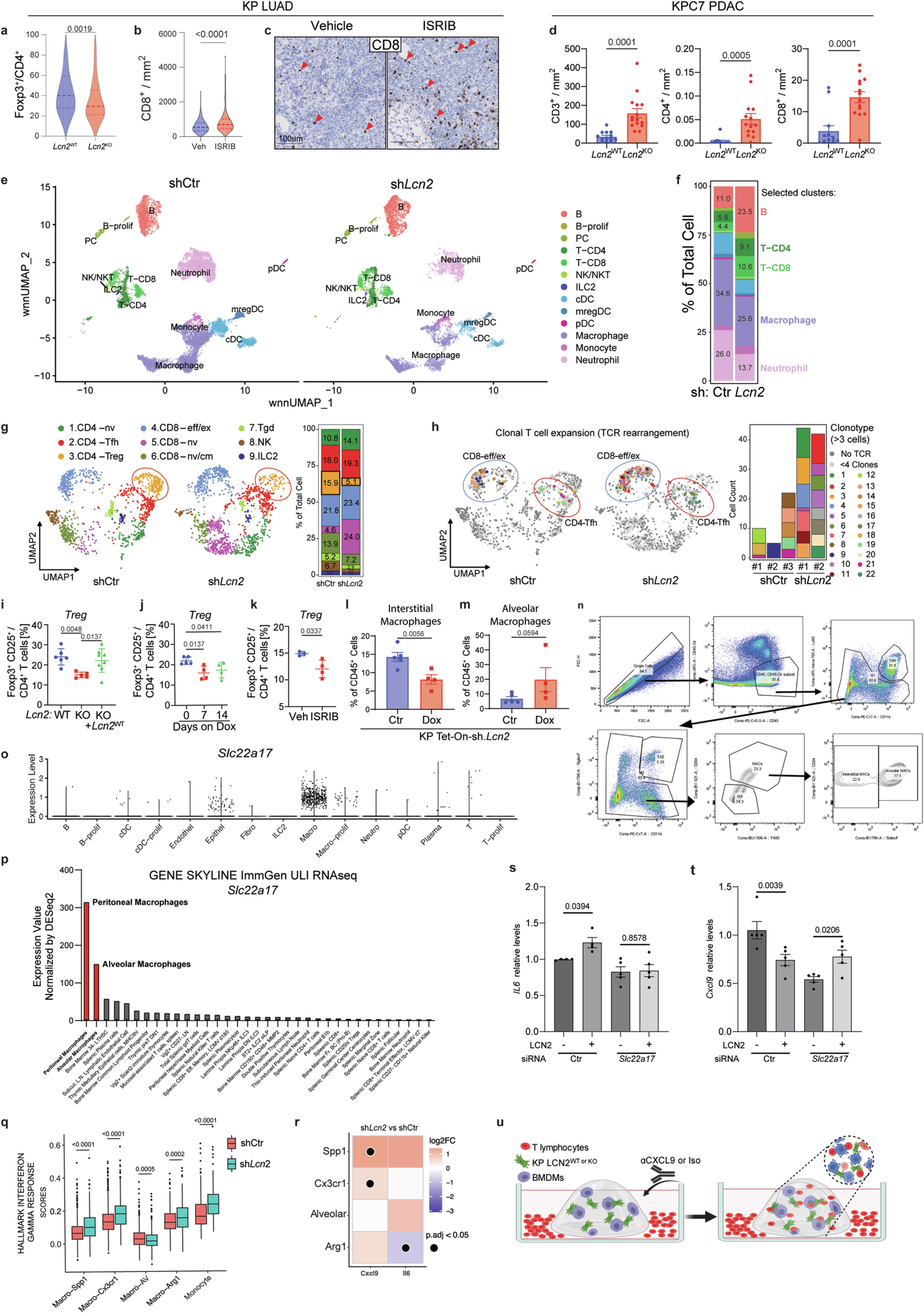
LCN2 is Closely Related to Human Tumor Malignancy and Immunotherapy Response
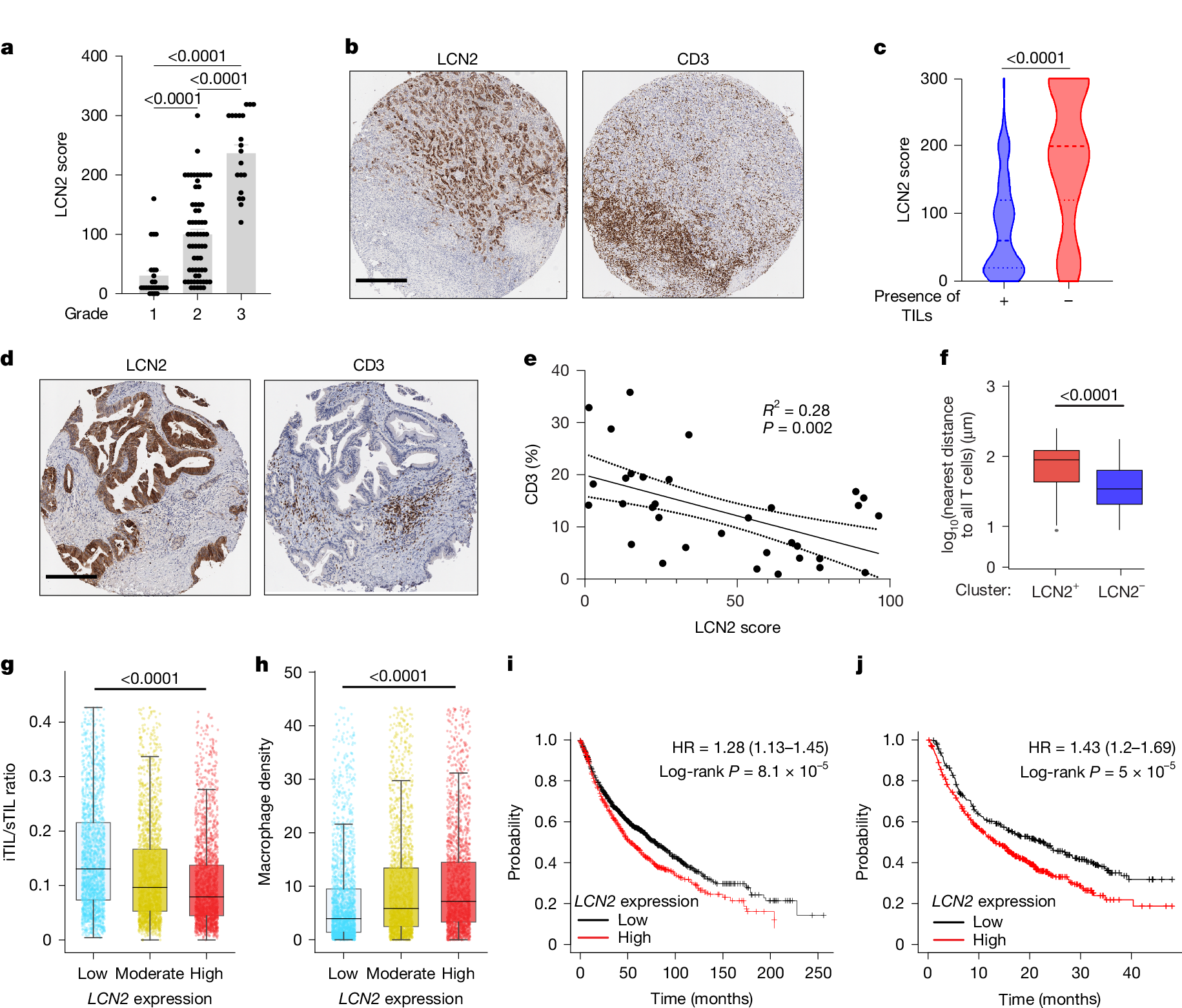
To clarify the clinical significance of LCN2, the study analyzed tumor tissue microarrays from 105 lung adenocarcinoma patients and found that LCN2 expression level was significantly positively correlated with tumor grade—the higher the tumor grade, the stronger the LCN2 expression. Moreover, areas with high LCN2 expression were often accompanied by reduced CD3+ T cell infiltration, and the positive rate of TILs in patients with high LCN2 expression was significantly lower than in patients with low expression. Similar negative correlation between LCN2 expression and CD3+ T cell infiltration was also observed in 33 pancreatic cancer patient samples. Single-cell spatial analysis showed that the spatial exclusion of T cells around LCN2-positive cells was significantly increased, suggesting that LCN2 exerts immune suppression effects locally in tumors.
TCGA pan-cancer analysis showed that LCN2 and ATF4 expression in immune-excluded tumors was significantly higher than in inflamed tumors. The ratio of intratumoral T cells to stromal T cells (iTIL/sTIL) in the LCN2 high-expression group was significantly reduced, while macrophage density was significantly increased, consistent with the results of mouse models. Survival analysis showed that lung cancer patients with high LCN2 expression had significantly shorter median survival than those with low expression, and a similar trend was observed in pancreatic cancer patients. Among cancer patients receiving immunotherapy, the survival of the LCN2 low-expression group was significantly longer than that of the high-expression group, suggesting that LCN2 can serve as a potential biomarker for predicting immunotherapy response.
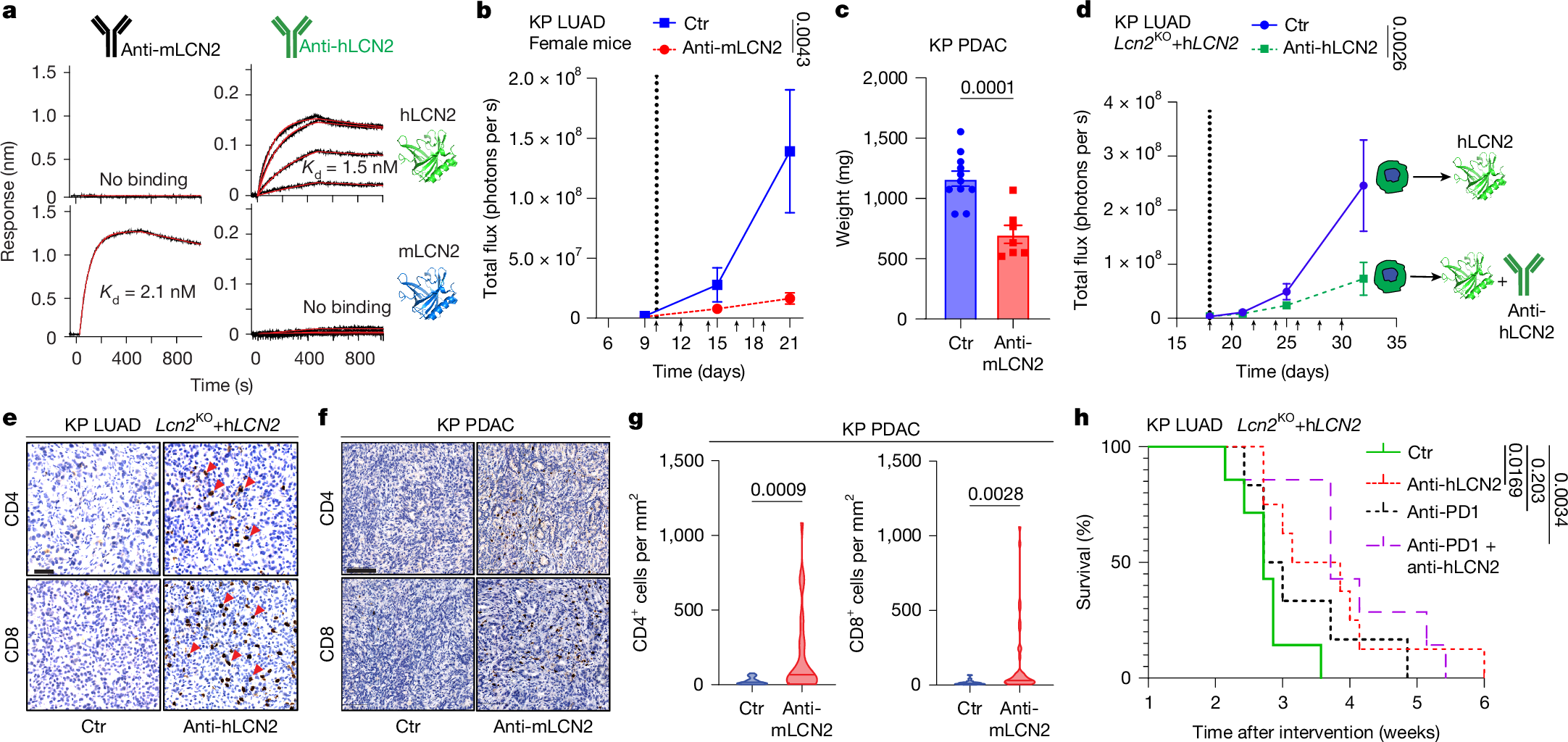
LCN2-Specific Antibodies Have Significant Anti-tumor Therapeutic Effects
Based on the secretory properties and clinical significance of LCN2, the study developed specific antibodies against mouse LCN2 (mLCN2) and human LCN2 (hLCN2). In an immunogenic tumor model (male primary tumor cells transplanted into female mice), anti-mLCN2 antibody treatment could significantly inhibit orthotopic tumor growth and prolong mouse survival. In a cold tumor model resistant to immune checkpoint inhibitors (male mice transplanted with KP lung cancer cells), anti-mLCN2 antibody could also effectively inhibit tumor growth without affecting the number of immune cells such as lymphocytes and neutrophils in mouse peripheral blood, with no obvious acute toxicity. In a pancreatic cancer orthotopic transplantation model, anti-mLCN2 antibody also showed significant anti-tumor effects.
To verify the therapeutic potential of anti-hLCN2 antibody, the study constructed an LCN2-knockout KP cell model expressing hLCN2 and found that anti-hLCN2 antibody could significantly inhibit tumor growth in this model, increase CD4+ and CD8+ T cell infiltration in tumors, and reduce Treg cell numbers. At the same time, anti-hLCN2 antibody could restore CXCL9 expression in macrophages, inhibit IL-6 expression, and reverse the LCN2-mediated immunosuppressive microenvironment. These results indicate that LCN2-specific antibodies can activate the body's anti-tumor immunity by blocking the immune suppression effect of LCN2, providing a new strategy for clinical treatment of immune-excluded solid tumors.
4. Research Summary
Through systematic in vitro and in vivo experiments, this study for the first time revealed the molecular mechanism by which the ISR-ATF4-LCN2 axis mediates tumor immune evasion: ATF4, as a key transcription factor downstream of ISR, directly binds to the LCN2 promoter and regulates its secretion. LCN2 secreted by tumor cells binds to SLC22A17 on the surface of macrophages, regulates macrophage phenotype, inhibits CXCL9 secretion, and reduces T cell infiltration, thereby promoting tumor immune evasion. The study also confirmed that LCN2 expression is closely related to the malignancy of human solid tumors, T cell infiltration, and immunotherapy response, and LCN2-specific antibodies have significant anti-tumor effects with good safety. This study not only fills the gap in the molecular mechanism by which ATF4 regulates tumor immune evasion, but also provides new therapeutic targets and biomarkers for lung cancer, pancreatic cancer, and other solid tumors that are resistant to immunotherapy, with important basic research value and clinical translation prospects.
References
Bossowski JP, Pillai R, Kilian J, Wong Lau A, Nakamura M, Rashidfarrokhi A, Hao Y, Li R, Wu K, Hattori T, Glasser E, Koide A, Wang L, Moreira AL, Hajdu C, Rajalingam S, LeBoeuf SE, Le H, Lee S, Oh JW, Joe C, Kim H, Ock CY, Lee SH, Wang H, Patel AAH, Sayin VI, Tsirigos A, Wong KK, Koralov SB, Pende M, Sánchez-Rivera FJ, Simeone DM, Zervantonakis IK, Koide S, Papagiannakopoulos T. The integrated stress response promotes immune evasion through lipocalin 2. Nature. 2026 Feb 18. doi: 10.1038/s41586-026-10143-0. Epub ahead of print. PMID: 41708864.