Literature Sharing: Combined Multiplex Immunofluorescence and Traditional Histology Imaging for Cancer Biomarker Discovery
1. Research Background
Precise analysis of the tumor microenvironment is the core foundation for cancer diagnosis, prognosis assessment, and treatment decision-making. However, traditional tissue imaging techniques have obvious limitations: single immunofluorescence (IF) imaging can detect multiple molecular markers but lacks histological context, making it difficult to accurately localize cell types and distributions. Hematoxylin-Eosin (H&E) staining, as the gold standard for pathological diagnosis, clearly presents tissue morphology but cannot provide molecular-level immune characteristic information. Existing multiplex imaging techniques either have complex processes and are time-consuming, or have issues such as fluorescence crosstalk and insufficient reagent stability, making it difficult to achieve simultaneous precise detection of molecular markers + tissue morphology.
To address this pain point, the research team developed a whole-slide imaging platform that combines H&E morphological knowledge with high-plex molecular information to achieve multi-dimensional detection of the same cells. Colorectal cancer (CRC), as a common malignant tumor, relies on tumor intrinsic characteristics and immune infiltration levels for prognosis assessment, but traditional immune scoring has limitations such as single parameters and limited predictive efficacy. This study aims to verify the reliability and reproducibility of Orion technology and develop a more precise colorectal cancer prognosis assessment model based on this technology, providing experimental basis for clinical translation.
2. Research Methods
This study focuses on the verification, optimization, and clinical application of Orion multimodal tissue imaging technology, covering three core aspects: technical system construction and verification, experimental sample and data collection, and data analysis and model construction.
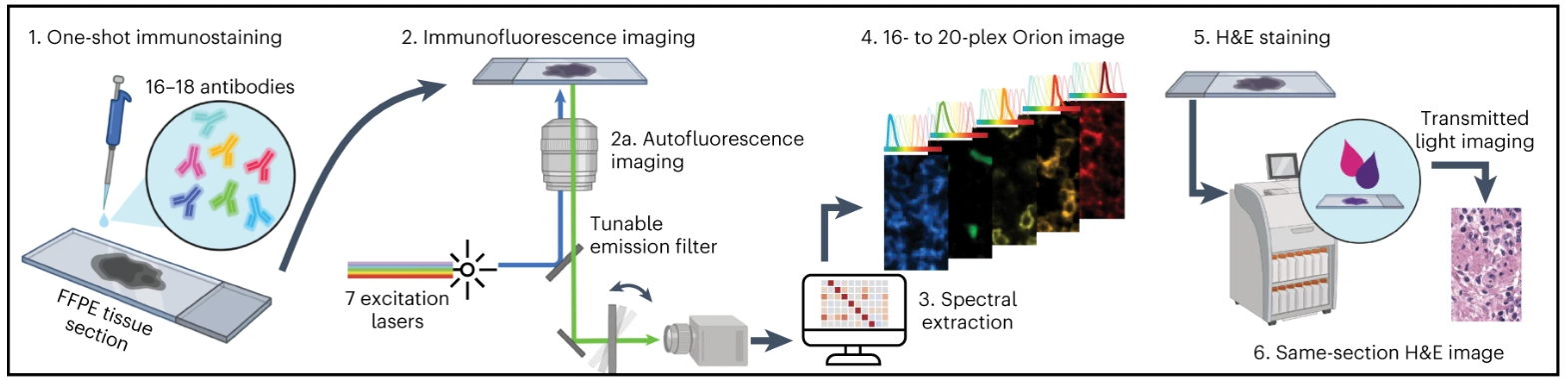
The experimental design and core process of multiplex immunofluorescence staining revolve around the adaptability, feasibility, and reproducibility of Orion technology. First, we designed and constructed an antibody panel suitable for studying epithelial tumors and their adjacent normal tissue microenvironments, covering immune cell markers, epithelial cell markers, endothelial cell markers, and functional markers. At the same time, antibody verification was completed by comparing with traditional immunohistochemical staining results, detecting adjacent channel fluorescence crosstalk, and verifying the crosstalk suppression effect of spectral extraction technology, ensuring the crosstalk rate is reduced to below 1%. The core process of staining and imaging focuses on solving the compatibility issue between H&E and IF. First, prepare FFPE tissue sections, including tonsil, lung cancer, CRC tissue, and tissue microarray TMA, perform a single round of Orion multiplex IF staining with 16-18 ArgoFluor-conjugated primary antibodies and complete imaging, then use an industrial standard slide stainer for H&E staining, which is confirmed by pathologists to reach diagnostic quality. Finally, obtain whole-slide images through the Orion imaging system and combine with software to complete data analysis such as spectral extraction and cell segmentation. To verify the effectiveness, reproducibility, and practicality of the method, the experiment set up three core groups: tonsil and lung cancer tissue verification group for verifying antibody panel applicability, TMA group containing 30 normal/non-tumor disease tissues and 18 tumor tissues for verifying broad applicability of the method, and a cohort of 74 stage I-IV CRC resection specimens for prognosis marker screening and model construction, among which 6 specimens were repeated in two different laboratories to verify cross-laboratory reproducibility.
3. Results Analysis
Feasibility of Multiplex Fluorescence Imaging and Channel Crosstalk
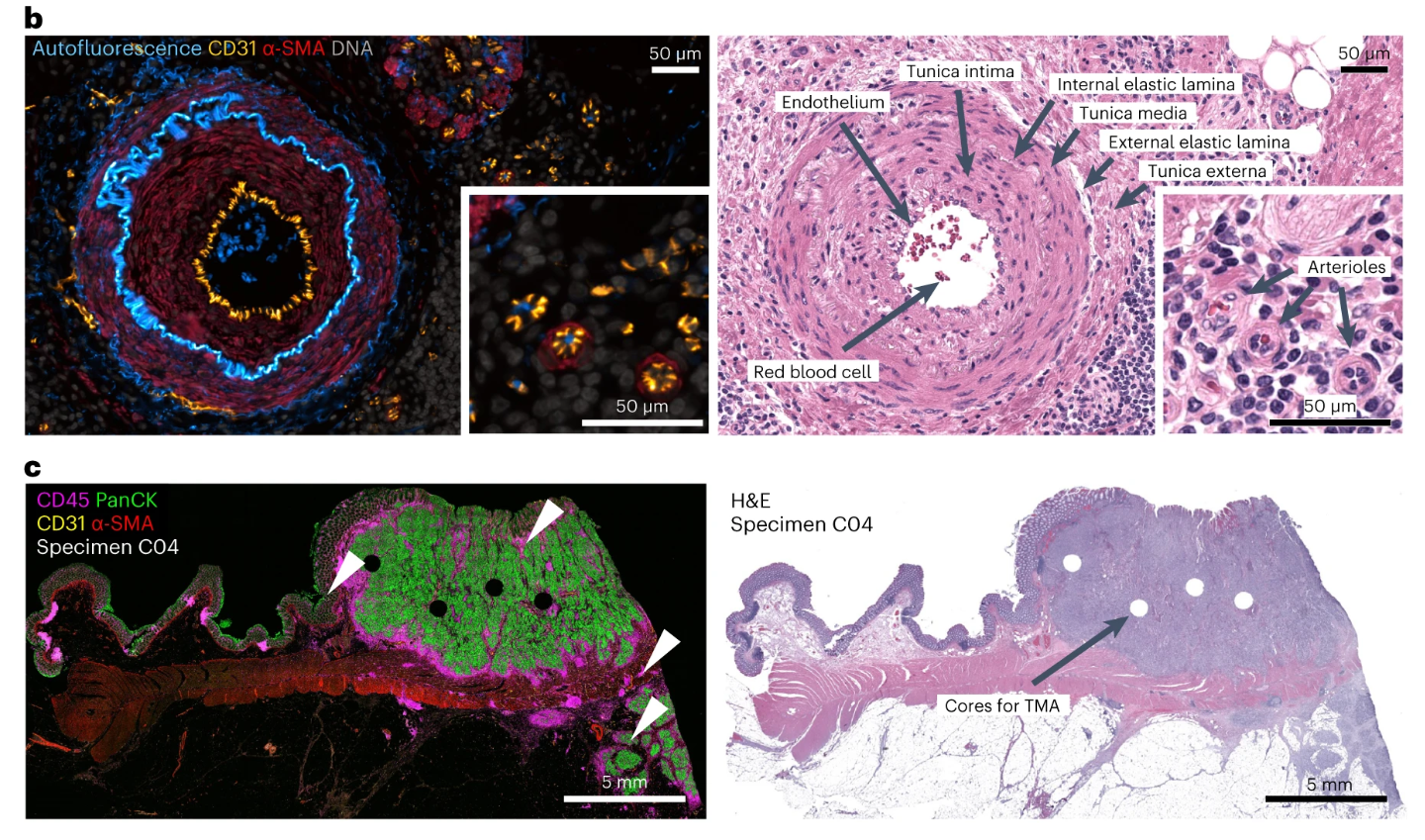
The study systematically verified the basic performance of Orion multiplex fluorescence imaging technology from different dimensions, providing core technical support for the subsequent experiments. The overlay imaging of the autofluorescence channel and IF antibody channel in the figure not only clearly presents the distribution characteristics of natural fluorescent structures such as connective tissue and blood vessels in the tissue, but also effectively highlights the specific fluorescence signals of target biomarkers, intuitively confirming the important role of the autofluorescence channel in improving biomarker signal-to-noise ratio and reducing background interference, ensuring the accuracy of subsequent biomarker detection.
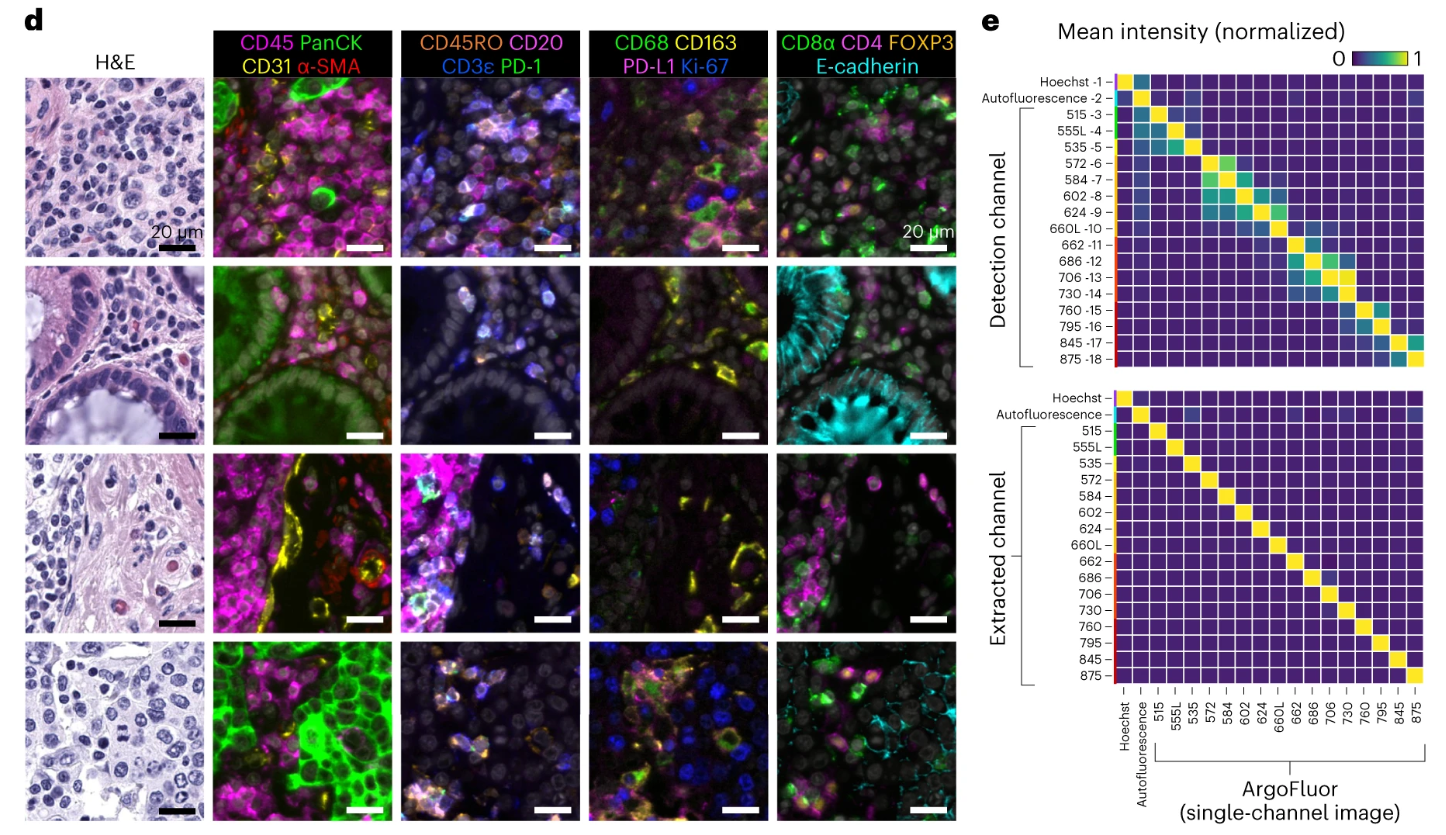
The high-resolution imaging details of the single image in Figure d clearly present the cell outline, nucleus, and fluorescence staining of target markers on the cell membrane, and can even distinguish subcellular-level fluorescence distribution, fully meeting the needs of subsequent refined analyses such as cell typing and subcellular localization. Figure e clearly demonstrates the significant effect of spectral extraction technology through quantitative analysis. In the experiment, tonsil tissue was labeled with a single antibody to detect crosstalk in adjacent fluorescence channels. The results showed that without spectral extraction, the average crosstalk in adjacent channels was approximately 35%, which could easily lead to confusion of fluorescence signals from different antibodies. After spectral extraction processing, the crosstalk rate was reduced to <1%, successfully solving the core technical pain point of channel crosstalk in multiplex fluorescence imaging, and providing reliable technical guarantee for single-round 18-20 plex and cyclic 32 plex high-plex imaging, ensuring that the fluorescence signal of each antibody can be accurately identified and quantified.
Multiplex Fluorescence Imaging Display
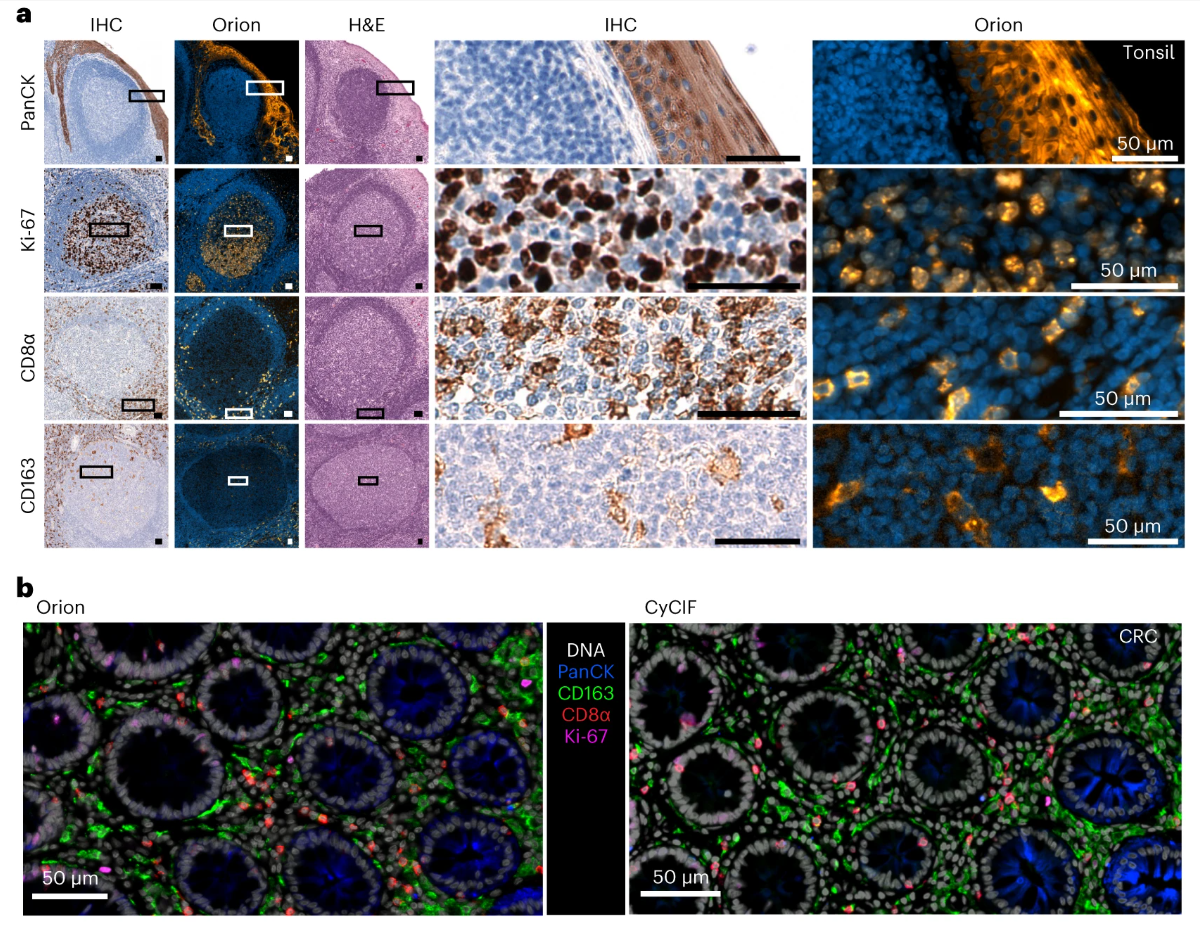
Through multi-dimensional comparison experiments, the reliability, accuracy, and practicality of Orion multiplex fluorescent antibody combinations were comprehensively verified. The study selected four core markers: pan-cytokeratin, Ki-67, CD8α, and CD163, and performed Orion multiplex IF and traditional IHC staining on the same tonsil, lung cancer, and CRC samples respectively. The results showed that the staining patterns of the two methods were highly similar, such as Ki-67 mainly localizing to the nucleus of actively proliferating cells, CD8α mainly distributed in immune infiltration areas, and pan-cytokeratin clearly marking tumor cell outlines, fully confirming the specificity of Orion antibody combinations. The quality of multiplex imaging is also not inferior to the mature CyCIF technology verified in the field, and the imaging method proposed in this paper has better resolution, which can better preserve cell detail information.
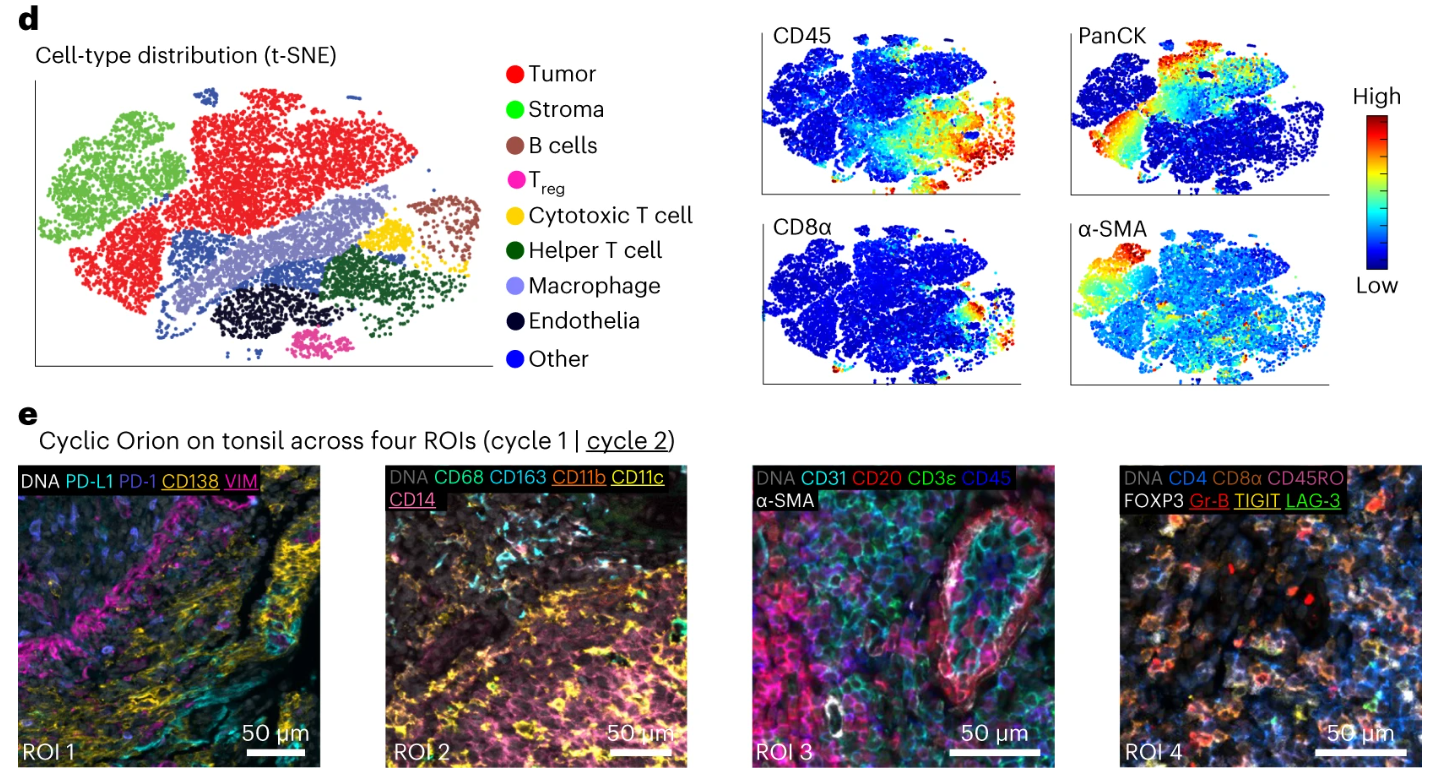
Based on 18-20 plex marker data obtained by Orion multiplex fluorescence imaging, the study performed t-SNE dimensionality reduction analysis, using algorithms to reduce high-dimensional fluorescence data to two-dimensional space, successfully clearly distinguishing expected cell populations such as tumor cells, immune cells, and stromal cells in CRC samples, with clear fluorescence characteristics for each cell population, proving that Orion multiplex fluorescence data can be effectively used for cell typing, providing a data basis for subsequent in-depth analysis at the cell level. Then, to verify the feasibility of cyclic Orion imaging, a method of hydrogen peroxide bleaching fluorophores was used to achieve 2 rounds of staining-imaging cycles, ultimately obtaining 32-plex imaging data for 29 antibodies. The experiment quantified that the bleaching step can reduce the fluorescence intensity of ArgoFluor fluorophores by >95%, with extremely low inter-round crosstalk, avoiding interference of previous round fluorescence signals on subsequent round imaging. At the same time, after 2 rounds of IF staining and bleaching treatment, the HE staining of the samples still maintained diagnostic quality, allowing clear identification of tissue morphology and cell structure, confirming that cyclic imaging can effectively increase the number of plexes without affecting subsequent histological analysis, providing a feasible solution for higher plex multiplex fluorescence imaging.
Joint Analysis of IF and HE Images
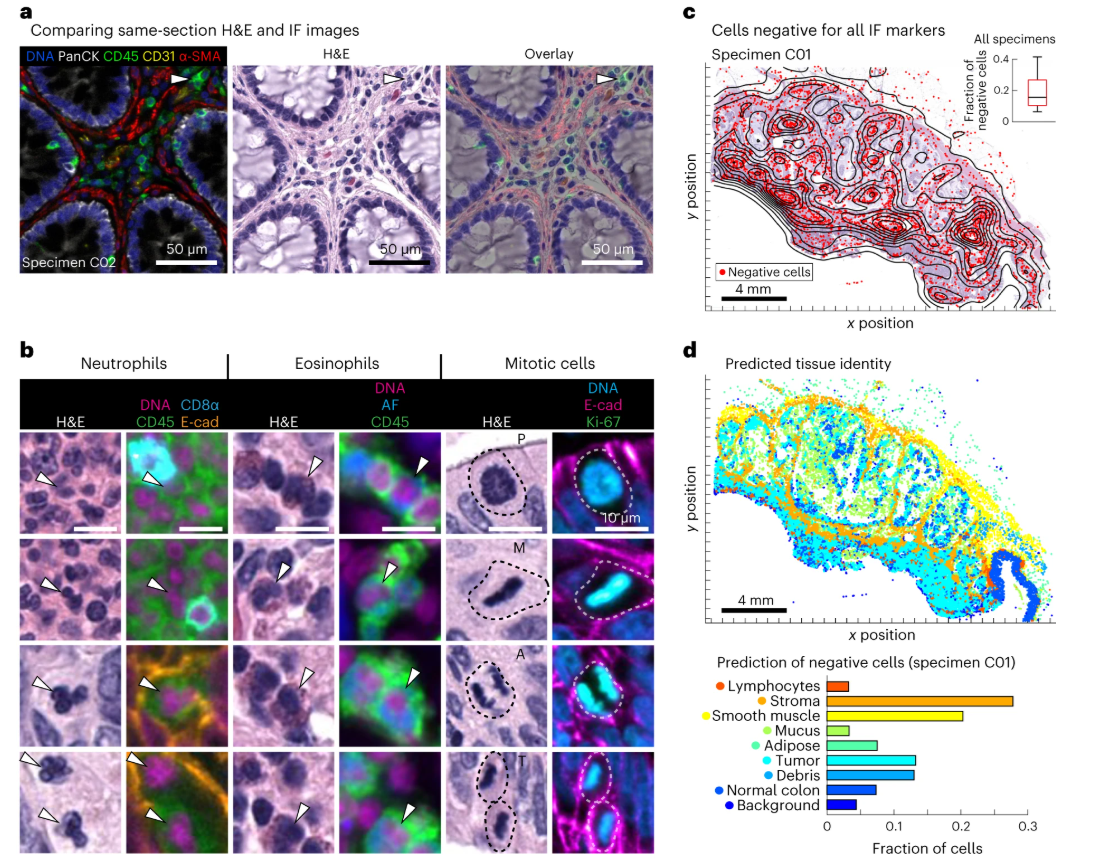
Through multiple comparison experiments, the complementarity of the two imaging techniques was fully verified, effectively solving the limitations of single imaging techniques and significantly improving the accuracy of cell typing and tissue structure identification. Figure a selects CRC samples for direct comparison of IF and HE images. IF imaging can clearly distinguish immune cell subsets with similar morphology and difficult to distinguish in HE images, such as CD4+ T cells, CD8+ T cells, and B cells, through fluorescent labeling of immune markers such as CD4, CD8, and CD20. These cells all appear as round lymphocytes in HE staining and cannot be distinguished by morphology, while IF can accurately identify them through specific fluorescent signals, highlighting the unique advantage of IF in cell molecular typing. Figure b focuses on the morphological advantages of HE staining. HE staining can clearly identify cell types without corresponding specific markers in IF, such as eosinophils and neutrophils, and can accurately distinguish various stages of cell mitosis. It can also clearly present acellular tissue components such as basement membranes and mucus pools. This information cannot be obtained through IF imaging, effectively compensating for the shortcomings of IF in morphological analysis.
Prognostic Model Construction Based on Orion Multiplex IF Data
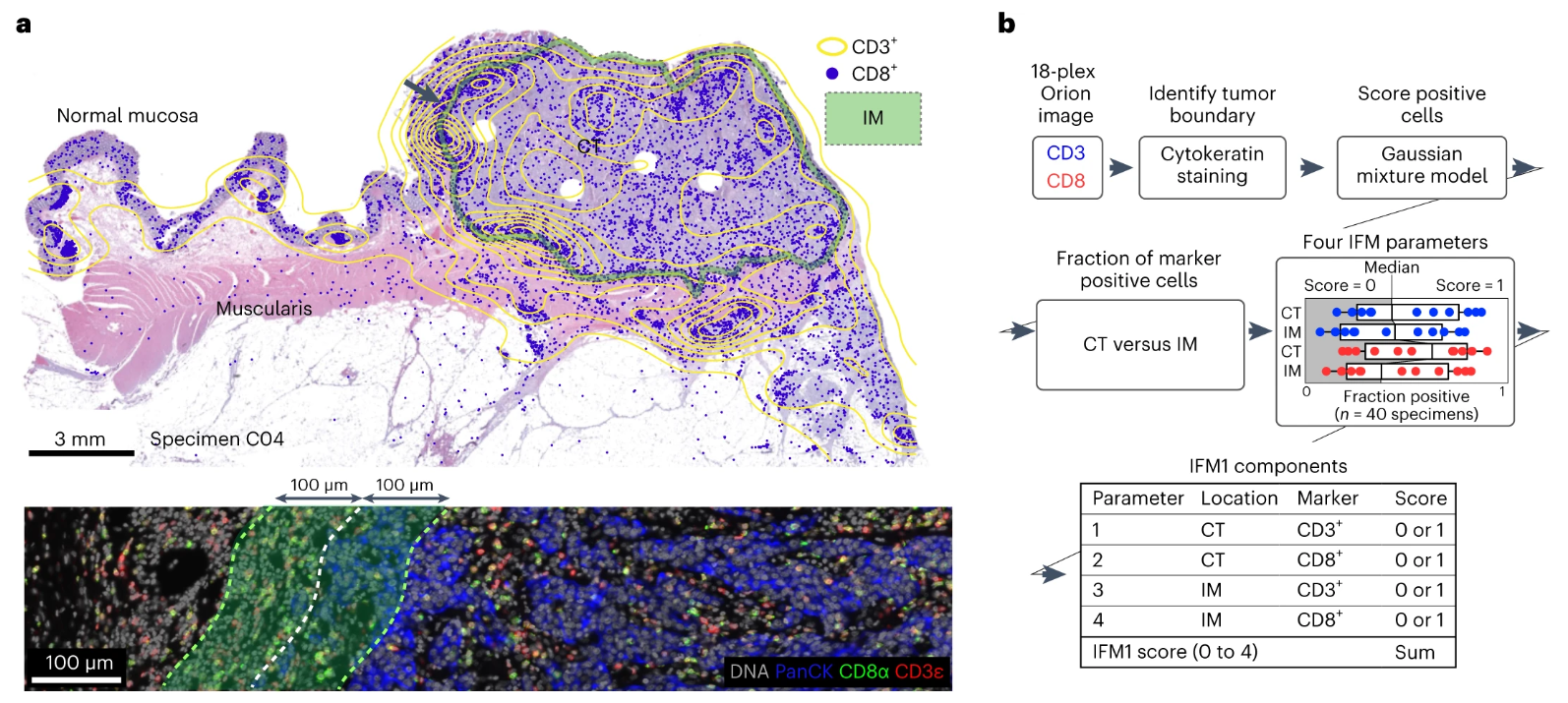
The study used fluorescence images of immune markers such as CD3 and CD8 obtained by Orion multiplex IF imaging, combined with autofluorescence channel and HE staining images, to clearly locate the boundary between tumor tissue and surrounding stroma, and precisely divide two key regions: CT (tumor center) and IM (tumor invasive margin, defined as tumor boundary ±100μm). Among them, the tumor center region is mainly composed of tumor cells with less immune cell infiltration, while the invasive margin region is the junction zone between tumor cells and immune cells with more abundant immune cell infiltration. This precise regional division provides a reliable spatial basis for subsequent region-specific cell counting, ensuring the accuracy and relevance of counting results. Figure b details the construction process of the IFM1 model. First, a Gaussian mixture model is used to analyze the fluorescence signals of CD3+ and CD8+ cells in Orion multiplex IF data to determine the positive thresholds for the two cell types, avoiding counting deviations caused by subjective judgment. Subsequently, the proportions of CD3+ and CD8+ positive cells in the CT region and IM region are counted separately, and 0-1 points are assigned according to the proportion levels. Orion multiplex IF data can efficiently replicate the prognostic prediction ability of Immunoscore, and compared with traditional methods, it has higher throughput and more objective quantification.
Novel Prognostic Marker Mining Based on Spatial-LDA
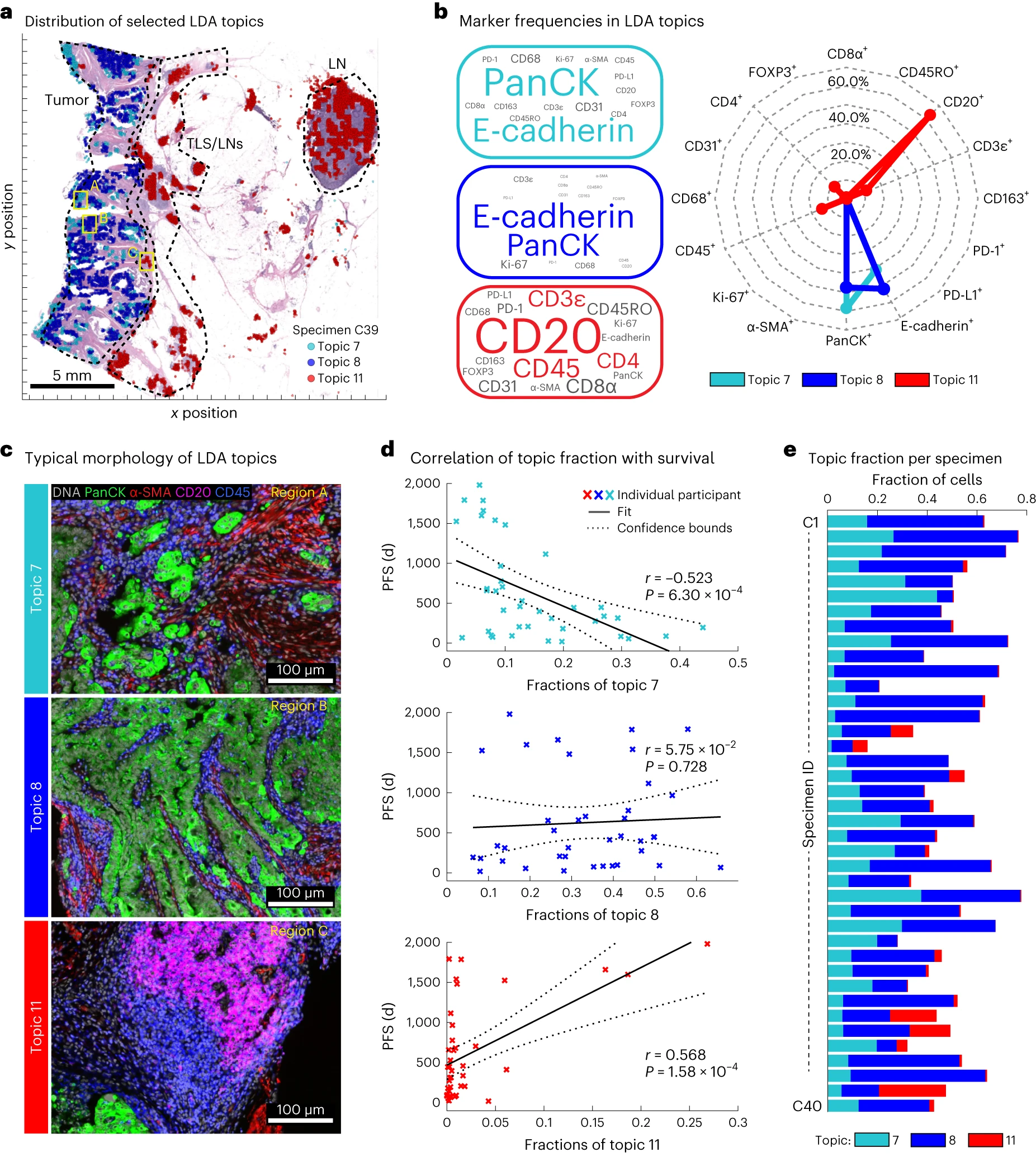
The authors used an unbiased Spatial-LDA algorithm to deeply mine Orion multiplex IF data, aiming to discover novel tumor intrinsic characteristics related to CRC prognosis, avoid marker omissions caused by traditional biased analysis, and provide new targets for building better prognostic models. Figure a shows the spatial distribution of 3 representative spatial features (topics), all overlaid with HE staining images to clarify tissue localization. Among them, topic7 is mainly distributed in the tumor center region, topic11 is mainly distributed in the tumor invasive margin and lymphoid aggregation regions, and topic8 is scattered in the tumor stroma region. It can be seen that different topics have significant differences in spatial localization in tumor tissues, proving that the Spatial-LDA algorithm can effectively distinguish different cell neighborhood structures and tissue region characteristics, and can capture spatial distribution differences that are difficult to detect by traditional analysis methods. Figure b clarifies the marker composition characteristics of each topic. Among them, topic7 is mainly composed of pan-cytokeratin and E-cadherin positive cells, with minimal contribution from immune cell markers, suggesting that it mainly reflects the characteristics of tumor cells themselves. Topic11 is mainly composed of CD20 positive cells, indicating its association with B cell infiltration. These spatial features provide new insights for understanding tumor heterogeneity and immune microenvironment.
4. Summary
This article introduces a multimodal tissue imaging method that combines single-round, high-throughput subcellular resolution immunofluorescence imaging, endogenous autofluorescence imaging, and subsequent HE staining imaging of the same cells. It has been verified that this multimodal tissue imaging has good reproducibility between different experimental platforms and has significant advantages for both human observers and machine learning models. Through Orion technology, the study proposes the combined use of IF and HE imaging, whose core value lies in using H&E tissue microanatomy knowledge to assist in interpreting molecular data, compensating for antibody type limitations, and being able to characterize acellular structures and distinguish cells with similar morphology but different functions. The Orion instrument used supports up to 18-plex simultaneous imaging, has been verified for applicability in 30 types of tissues, and can increase molecular channels through cyclic imaging or combination with CyCIF. In diagnostic applications, only 8-14 channels are needed to reduce costs, and H&E can identify cells that are difficult to distinguish by multiplex IF, while IF can assist in distinguishing immune cells, complementing each other and providing powerful tools for clinical diagnosis and scientific research. The 4 prognostic models constructed based on Orion technology are significantly superior to traditional immune scoring and can accurately predict the progression-free survival of colorectal cancer patients. At the same time, new spatial features related to prognosis were mined through Spatial-LDA, providing new ideas and markers for the prognosis assessment of colorectal cancer.
References
Lin JR, Chen YA, Campton D, Cooper J, Coy S, Yapp C, Tefft JB, McCarty E, Ligon KL, Rodig SJ, Reese S, George T, Santagata S, Sorger PK. High-plex immunofluorescence imaging and traditional histology of the same tissue section for discovering image-based biomarkers. Nat Cancer. 2023 Jul;4(7):1036-1052. doi: 10.1038/s43018-023-00576-1. Epub 2023 Jun 22. PMID: 37349501; PMCID: PMC10368530.
EnkiLife mIHC TSA Kits
Product | Catalog Number |
|---|---|
TSA Six-Label Seven-Color Multiplex Immunohistochemistry Kit | |
TSA Five-Label Six-Color Multiplex Immunohistochemistry Kit | |
TSA Four-Label Five-Color Multiplex Immunohistochemistry Kit | |
TSA Three-Label Four-Color Multiplex Immunohistochemistry Kit | |
TSA Two-Label Three-Color Multiplex Immunohistochemistry Kit |
