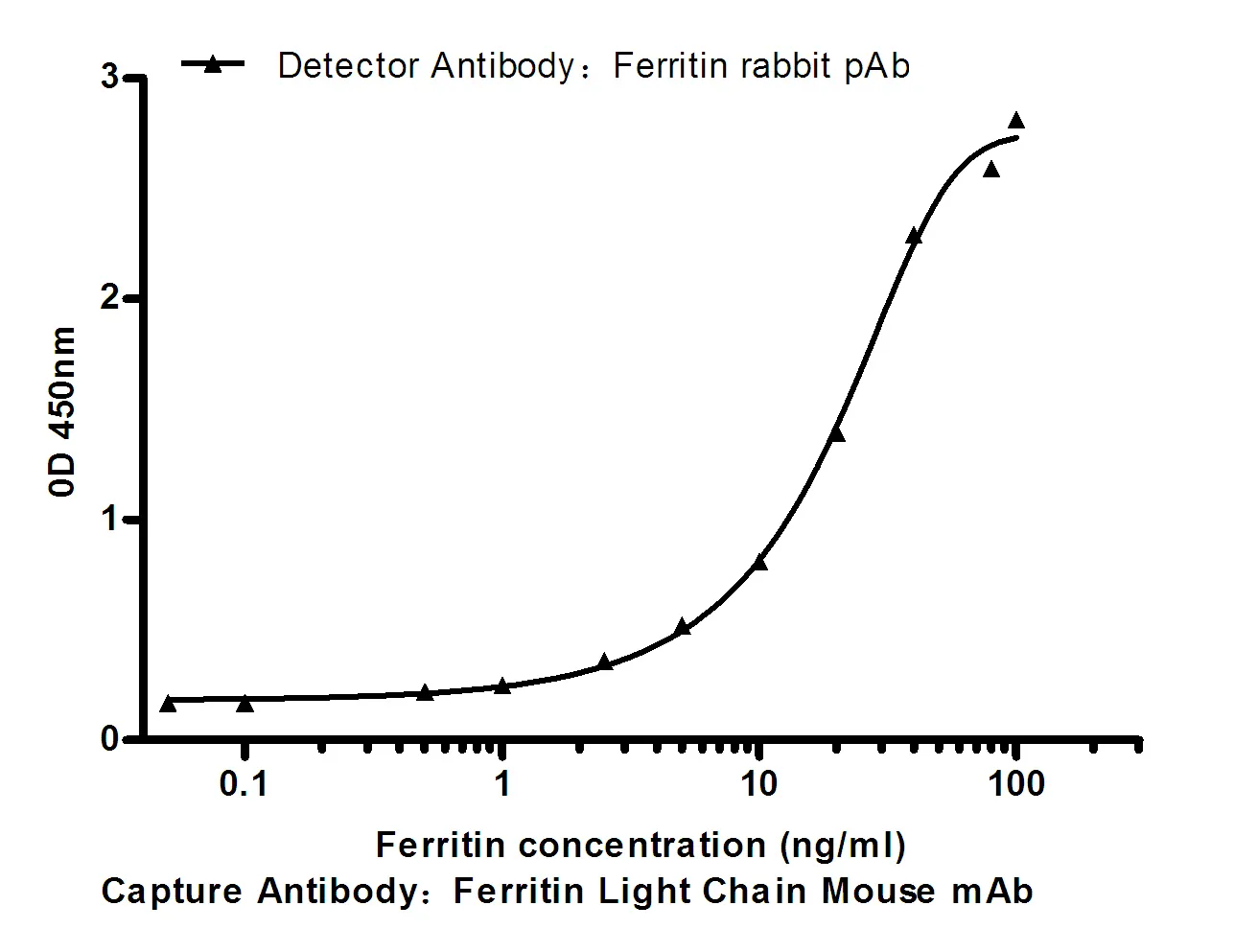
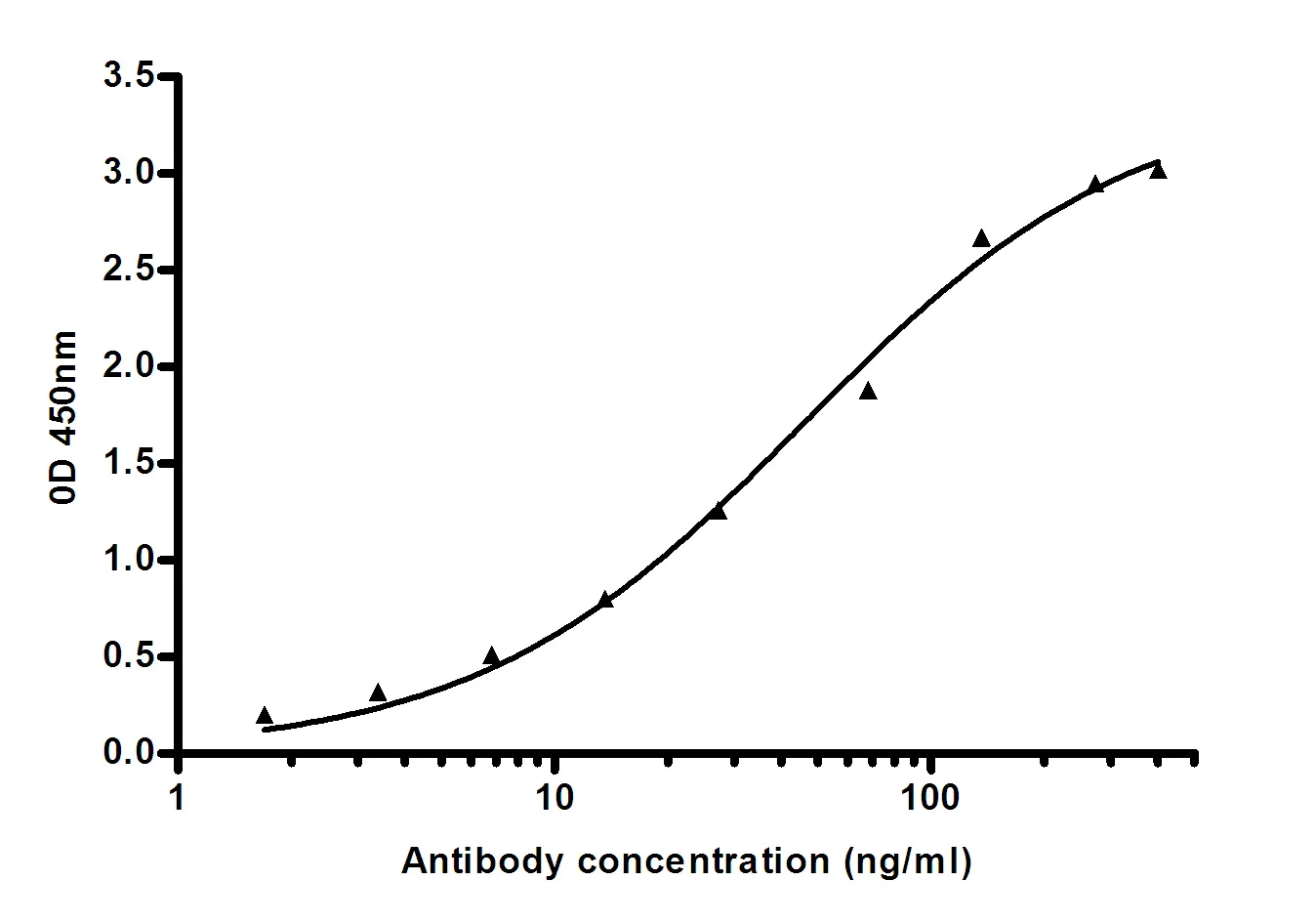
Summary
Performance
Immunogen
Application
Background
The protein encoded by this gene is a bifunctional enzyme that initiates and regulates the biosynthesis of N-acetylneuraminic acid (NeuAc), a precursor of sialic acids. It is a rate-limiting enzyme in the sialic acid biosynthetic pathway. Sialic acid modification of cell surface molecules is crucial for their function in many biologic processes, including cell adhesion and signal transduction. Differential sialylation of cell surface molecules is also implicated in the tumorigenicity and metastatic behavior of malignant cells. Mutations in this gene are associated with sialuria, autosomal recessive inclusion body myopathy, and Nonaka myopathy. Alternative splicing of this gene results in transcript variants encoding different isoforms. [provided by RefSeq, Jul 2008],catalytic activity:ATP + N-acyl-D-mannosamine = ADP + N-acyl-D-mannosamine 6-phosphate.,catalytic activity:UDP-N-acetyl-D-glucosamine = UDP-N-acetyl-D-mannosamine.,disease:Defects in GNE are a cause of sialuria [MIM:269921]; also known as sialuria French type. In sialuria, free sialic acid accumulates in the cytoplasm and gram quantities of neuraminic acid are secreted in the urine. The metabolic defect involves lack of feedback inhibition of UDP-GlcNAc 2-epimerase by CMP-Neu5Ac, resulting in constitutive overproduction of free Neu5Ac. Clinical features include variable degrees of developmental delay, coarse facial features and hepatomegaly. Sialuria inheritance is autosomal dominant.,disease:Defects in GNE are the cause of inclusion body myopathy type 2 (IBM2) [MIM:600737]. Hereditary inclusion body myopathies are a group of neuromuscular disorders characterized by adult onset, slowly progressive distal and proximal weakness and a typical muscle pathology including rimmed vacuoles and filamentous inclusions. IBM2 is an autosomal recessive disorder affecting mainly leg muscles, but with an unusual distribution that spares the quadriceps as also observed in Nonaka myopathy.,disease:Defects in GNE are the cause of Nonaka myopathy (NM) [MIM:605820]; also known as distal myopathy with rimmed vacuoles (DMRV). NM is an autosomal recessive muscular disorder, allelic to inclusion body myopathy 2. It is characterized by weakness of the anterior compartment of the lower limbs with onset in early adulthood, and sparing of the quadriceps muscles. As the inclusion body myopathy, NM is histologically characterized by the presence of numerous rimmed vacuoles without inflammatory changes in muscle specimens.,enzyme regulation:Allosterically regulated (Probable); feedback inhibited by cytidine monophosphate-N-acetylneuraminic acid (CMP-Neu5Ac), the end product of neuraminic acid biosynthesis. Activity is dependent on oligomerization. The monomer is inactive, whereas the dimer catalyzes only the phosphorylation of N-acetylmannosamine; the hexamer is fully active for both enzyme activities (By similarity). Up-regulated after PKC-dependent phosphorylation.,function:Regulates and initiates biosynthesis of N-acetylneuraminic acid (NeuAc), a precursor of sialic acids. Plays an essential role in early development (By similarity). Required for normal sialylation in hematopoietic cells. Sialylation is implicated in cell adhesion, signal transduction, tumorigenicity and metastatic behavior of malignant cells.,pathway:Amino-sugar metabolism; N-acetylneuraminic acid biosynthesis.,PTM:Phosphorylated by PKC.,similarity:In the C-terminal section; belongs to the ROK (nagC/xylR) family.,similarity:In the N-terminal section; belongs to the UDP-N-acetylglucosamine 2-epimerase family.,subunit:Homodimer and homohexamer.,tissue specificity:Highest expression in liver and placenta. Also found in heart, brain, lung, kidney, skeletal muscle and pancreas.,
Research Area
Amino sugar and nucleotide sugar metabolism;